INTRODUCTION
Tumors of the pancreas are divided into exocrine and endocrine tumors. Ductal carcinoma is the most common exocrine tumor of the pancreas and is subdivided into adenocarcinoma and squamous cell carcinoma. Adenocarcinoma, the most common subtype, comprises 80% of all malignant tumors of the pancreas,1,2 while primary squamous cell carcinoma is an extremely rare subtype of ductal carcinoma in the pancreas because the cell line does not exist in a normal pancreas.3,4 Consequently, if the presence of squamous cell carcinoma is confirmed in a biopsy, the possibility of metastatic disease must be considered first, and an extensive search for the primary lesion should be undertaken.5,6
Recently, we confirmed a case of synchronous esophageal cancer with pancreatic metastasis and hepatocellular carcinoma (HCC). This is reported here along with a literature review.
CASE REPORT
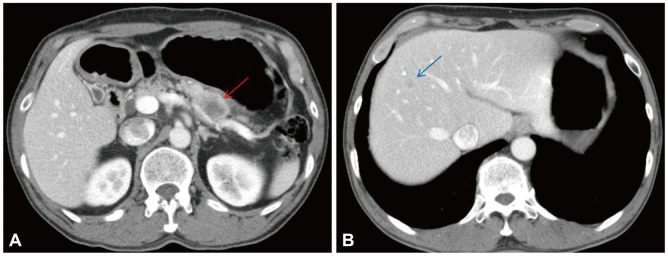
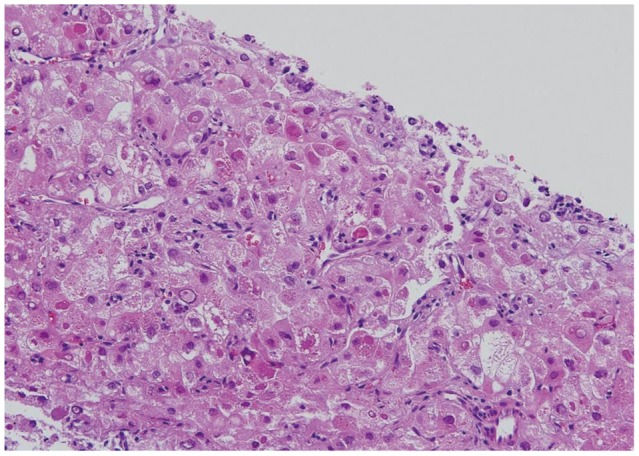
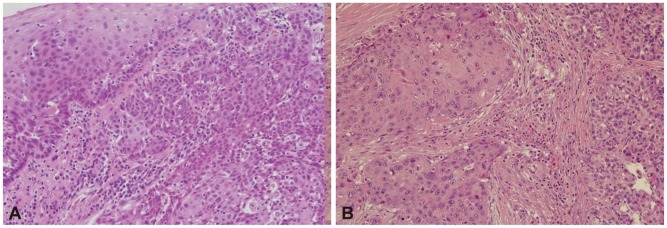
A 58-year-old male with a 1-month history of abdominal bloating and indigestion visited our hospital for esophagogastroduodenoscopy. This detected a 3У4-cm erythematous lesion with an irregular nodular surface extending between 38 and 41 cm from the incisors in the 9 to 1 o,clock position (Fig. 1). With Iodine staining, it was shown as an iodine-void area with a well-defined boundary. A biopsy revealed squamous cell carcinoma, and he was admitted for further evaluation and management of this esophageal cancer. He was an alcoholic who had been drinking one bottle of Soju daily for the past 30 years and had a 30-pack-year smoking history. He was a hepatitis B carrier without regular follow-up. At admission, the patient's vitals were stable and he was alert mentally. No abnormalities were detected in the physical examination. The peripheral blood tests were normal, with hemoglobin 13.6 g/dL, white blood cell count 7,330/mm3, platelets 238,000/mm3, as were the blood chemistry and electrolytes. Additional studies for hepatitis B showed hepatitis B e antigen (-) and anti-hepatitis B e antibody (+), and no virus was detected with hepatitis B virus real-time polymerase chain reaction. The results for tumor markers were as follows: carcinoembryonic antigen 2.39 ng/mL (reference range, <4.1), CA19-9 2 U/mL (reference range, <37), and alpha-fetoprotein (AFP) 8.58 ng/mL (reference range, <4.0). Abdominal computed tomography (CT) showed a poorly demarcated and poorly enhancing mass at the pancreatic tail (Fig. 2A). Magnetic resonance imaging (MRI) showed that the pancreatic mass appeared poorly demarcated and hyperintense on T2-weighted image and hypointense on T1-weighted image. Dynamic enhancing MRI showed that the mass was poorly enhanced without rim enhancement. So we were highly suspicious that the pancreatic mass was a primary adenocarcinoma rather than metastasis, because of tumor character and extremely low incidence of pancreatic metastasis from esophageal cancer. CT and MRI showed 1.2 cm-sized single nodule in the liver (Fig. 2B). MRI showed that the hepatic mass was hypointense on T1-weighted image and hyperintense on T2-weighted image, and poorly enhanced on dynamic enhancing study. However, serum AFP was elevated (8.52 ng/mL) and we had to undergo ultrasound-guided liver biopsy. The hepatic mass was confirmed as a primary HCC based on the loss of ductular reaction and CD34 positivity (Fig. 3). The patient underwent surgery for the esophagus and pancreas lesions and radiofrequency ablation for the hepatic cancer. Histologically, the partial esophagectomy and distal pancreatectomy specimens showed squamous cell carcinoma and local lymph node invasion and the patient was diagnosed with stage IV esophageal cancer with pancreatic metastasis (Fig. 4). In the esophagus, the tumor invaded to submucosal layer with lymphatic invasion and metastasis to 3 of 31 regional lymph nodes. In the pancreas, the size was 5У4.5У4 cm with clean resection margins but there was lymphatic invasion involving 2 of 20 regional lymph nodes. The patient had received 5-fluorouracil/cisplatin combination therapy six times for 4 months after the surgery. He has been followed up without any evidence of recurrence on abdomen CT and gastroscopy until this month.
DISCUSSION
Epithelial tumors of the pancreas are divided into exocrine tumors, which are derived from ductal and acinar cells, and endocrine tumors, which are derived from islet cells.1,2 Most malignant tumors in the pancreas are primary, and approximately 80% are adenocarcinomas. The rest include metastatic lesions, squamous cell carcinoma, small cell carcinoma, and acinar cell carcinoma.7 Primary squamous cell carcinoma of the pancreas is extremely rare, accounting for only 0.7% of 1,300 pancreatic cancer in a 1992 report from Japan because the normal pancreas does not contain squamous epithelium.8 Typically, primary squamous cell carcinoma of the pancreas is confirmed only after other primary sources have been excluded using diagnostic tests like CT of the head and chest, gastrointestinal endoscopy, a pelvic exam with a pap smear, and a thorough examination of the skin. Once there is no evidence of a primary lesion elsewhere, the diagnosis of primary pancreatic cancer can be made.5,6 Isolated pancreatic metastases from nonpancreatic primary tumors are also very rare, accounting for about 2% of all pancreatic tumors. Autopsy studies of cancer patients with pancreatic metastasis have shown that the lungs and liver are the most common sites of primary lesions, followed by the colon and kidney. Primary esophageal cancer compromises only 0% to 4.8% of pancreatic metastasis, and there has been no reported case in South Korea.8-10 Generally, it is difficult to distinguish a solitary pancreatic metastasis from a primary cancer and a double or triple primary pancreatic cancer is difficult to diagnose in almost all cases. A tissue diagnosis can be made before surgery with a fine needle aspiration biopsy, guided by ultrasonography, CT, or another modality. In the case of a pancreatic mass, resectability should be considered ahead of a preoperative biopsy.
In our case, both primary pancreatic cancer and pancreatic metastasis from the esophageal cancer or HCC were possible. But the pancreatic mass was considered as primary cancer on abdomen CT and MRI. So we decided surgical resection because the tumors were thought to be triple primary cancer arising from liver, esophagus, and pancreas. After the operation, the pancreatic mass was unexpectedly confirmed as metastasis from the esophageal cancer.
In the treatment of cancer patients with an isolated distant organ metastasis and no widespread disease, resection of the metastasis is beneficial for some types of tumor. Hiotis et al.11 reported that the resection of pancreatic metastatic lesions in 16 patients with different primary tumors led to a longer disease-free interval and improved overall survival. In addition, the surgical resection of solitary metastatic lesions from colorectal cancer to the pancreas increases patient survival.10 Furthermore, the resection of isolated extrahepatic recurrences of HCC prolonged the survival in selected patients.12 A review of the surgical outcomes of reported cases, including our patient, indicated that the surgical mortality rate in pancreatic surgery is less than 5%.13 In our case, surgical resection was reasonable for treatment and obtaining a definite diagnosis, as well as for improving patient survival, although there are few guidelines for managing cases such as ours.
However, these previous reports are based on metastatic lesions that developed after the diagnosis of well-controlled primary tumors. There are some limitations to applying these reported cases to our case, in which a primary lesion and an isolated metastatic lesion were found simultaneously and the metastatic lesion was considered as primary tumor on imaging studies. Further observations of the prognosis are required in cases diagnosed with synchronous double primary cancer with an isolated metastatic lesion. We reported this case because isolated pancreatic metastasis of esophageal cancer is rare, and careful treatment decisions are required. Moreover, established guidelines for treating double primary cancer with an isolated metastatic lesion are needed.