INTRODUCTION
Bile duct cancer (BDC) is an uncommon malignant tumor that may arise anywhere that biliary epithelium is present from the ampulla of Vater to the smallest intrahepatic biliary radicles. BDCs are classified into three main categories: perihilar BDCs (also known as Klatskin's tumors), distal BDCs, and intrahepatic BDCs.1 Perihilar BDCs are further subclassified depending on the degree of the proximal tumor extension within the intrahepatic biliary radicles according to the classification proposed and later modified by Bismuth.2 Klatskin's tumors are the most common form of BDCs and account for approximately 70% of cases of BDCs. Distal and intrahepatic BDCs account for approximately 25% and 5% of cases, respectively.1
Complete resection with negative margins is the only treatment with the potential for cure, with 5-year survival rates of 20% to 40%.3,4 However, patients with cholangiocarcinoma (CC) usually present at an advanced stage, with more than 50% being unresectable at the time of diagnosis. Recently, two randomized controlled trials have shown a significant survival benefit in patients with unresectable CC treated with photodynamic therapy (PDT).5,6 One of these studies also showed a significant improvement in quality of life after PDT and stenting.6
PDT is based on the relatively specific accumulation of photosensitizers, such as porphyrins, in dysplastic or malignant cells. CC cell lines have shown favorable cellular uptake kinetics for sodium porfimer and excellent phototoxic cell damage in response to PDT in vitro and in vivo in a human CC xenograft model in nude mice.7,8 In this paper, we review recent advances of PDT in BDCs.
SPECIFIC FEATURES OF TUMOR BIOLOGY OF BDCS ARE SUITABLE FOR PDT
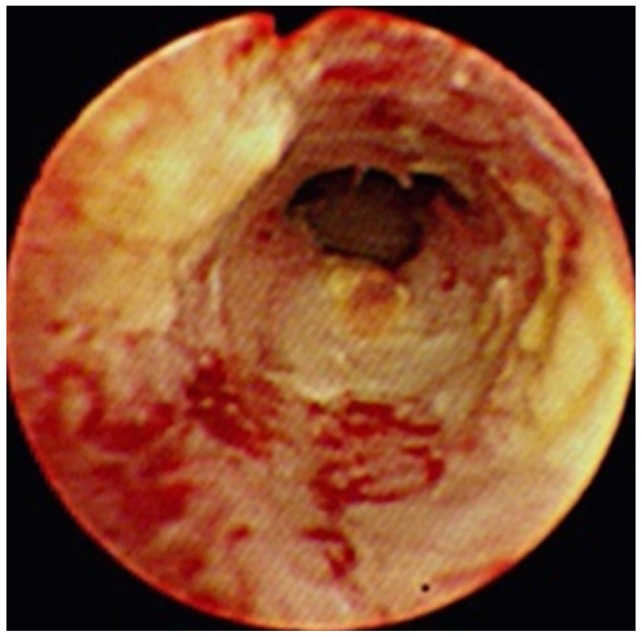
CC is characterized by a usually slow growth rate and a low propensity for metastasis. The most common form in BDCs is a highly desmoplastic cancer with a growth pattern characterized by periductal extension and infiltration (Fig. 1).9 This form of CC often obstructs bile ducts and encases blood vessels strangulating these structures mechanically and disrupting bile and blood flow, respectively. Because these cancers involve both intrahepatic and extrahepatic bile ducts, they will simply be referred to as ductal CCs: however, given their predilection to occur at the bifurcation of the right and left hepatic duct, others have referred to these cancers as perihilar CCs (Klatskin's tumor). In contrast, the other principal form of this disease grows as a mass lesion within the liver. This form of the disease will be referred to as intrahepatic CCs because they, in part, mimic hepatocelluar carcinomas. In addition to different growth pattern, current information suggests these two forms of CC may differ in their etiopathogenesis, risk factors, and perhaps molecular and genetic signatures.10
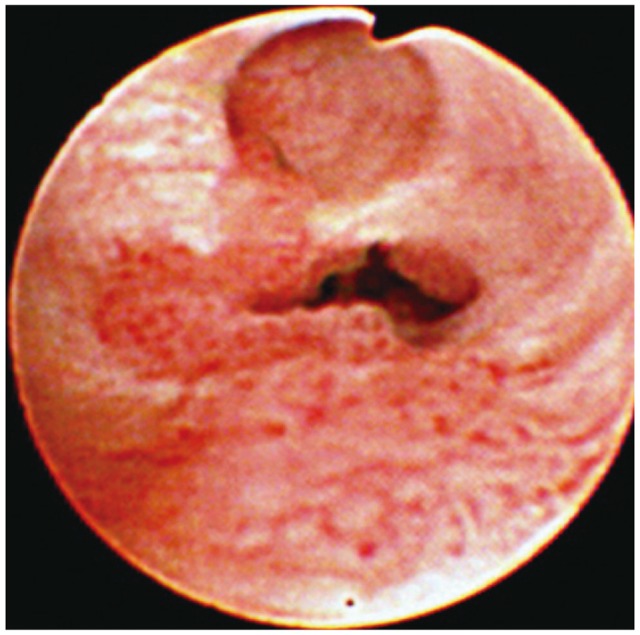
Although nearly all CCs are well-differentiated adenocarcinoma, their tumor biology is a little different from other gastrointestinal adenocarcinoma, especially in hilar bile duct cancer (HBDC). In general, hematogenous spread of HBDC is rare, whereas nodal metastases may be present in up to one-third of cases.11 Extensive subepithelial tumors spread beyond the gross tumor margin is common, and longitudinal tumors may extend 15 to 20 mm proximally and 5 to 10 mm distally, depending on tumor type.12,13 The papillary variant composes 10% of all CCs and grows primary as an intraluminal soft, polypoid tumor with a limited propensity for transmural growth (Fig. 2). Tumor multicentricity occurs more frequently with the papillary variant and may be reflective of a field change in the biliary epithelium.11,14,15 The nodular variant occurs most commonly in the upper and mid bile duct and generally presents as a fibrotic mass with intraductal projections (Fig. 3). The sclerosing variant comprises 70% of all tumors at the hilum and histologically appears as annular thickening of the bile duct wall with both longitudinal and radial tumor infiltrations.
WHAT ARE THE APPROPRIATE INDICATIONS OF PDT IN BDCS?
Neoadjuvant PDT for hilar CC showed efficacious tumor destruction confined to the superficial 4- to 4.5-mm depth of the bile duct tumor and high tumor selectivity in the resected bile duct specimens.16 This cannot eradicate the primary tumor when tumor invasion extends to a depth of 7 to 9 mm. Therefore the appropriate indications of PDT in BDCs may be as follows: 1) sclerosing variant without hematogenous metastasis regardless of nodal metastases; 2) superficial spreading type with the papillary variant; and 3) R0 and R1 residual tumor after resection. The mass-forming intrahepatic CCs, intraductal mass form of BDCs and cases of hematogenous metastases are not indicated for PDT.
RATIONAL OF LOCAL TUMOR ABLATION IN BDCS
About 80% of BDCs, especially perihilar CCs, are adenocarcinomas that exhibit predominantly a longitudinal growth pattern along the biliary tree, most highly desmoplastic tumors with infiltration of adjacent nerve plexus and lymphatics.9
Even with an aggressive surgical approach only 33% to 50% are resectable, and in only 28% are negative histological margins obtained.17
The majority of patients with tumor stenoses in the distal and middle part of the bile duct cholestasis can be relieved quickly by stenting. The role of palliative intervention is limited in proximal BDCs. Independent of the type of stricture, technically successful endoprosthesis placement is possible in 84% to 96% of these patients.18,19 A successful drainage (bilirubin decrease >30% to 50%) is only achieved, however, in 69% to 91% of Bismuth type I and II stenosis and in 15% to 73% of Bismuth type III and IV tumors.18,20 Although metal stent insertion improves occlusion rates and reduces the number of therapeutic interventions, median survival time is not ameliorated.21,22 Attempts to affect tumor growth are made with radiotherapy or chemotherapy. Until now, however, chemotherapy is unsatisfactory in terms of survival times although chemotherapy with gemcitabine and platinum achieved a modest improvement in survival compared to other regimens.23 Whether radiotherapy is able to improve survival is still on debate. The retrospective comparative study of palliative radiation therapy showed no significantly improved median survival time (300 days vs. 210 days) between endoscopic biliary stenting with or without external-beam radiotherapy and internal 192Ir brachytherapy.24 Recently, stereotatic body radiotherapy may be a new promising option but has to be evaluated further.25
A treatment modality for local ablation of the primary tumor could improve the outcome of curative as well as palliative therapies. Palliative brachytherapy with only 192-iridium (dose of 35 Gy in 1-cm distance) did not prolong median survival time (4.3 to 5 months),26 but when combined with external beam radiotherapy (30 Gy), resulted in median survival times of 10 to 10.5 months.21 Another modality for local tumor ablation of CC is PDT.
PHOTODYNAMIC EFFECT IN BDCS
The tumor-selective enrichment of porfimer has been confirmed in human BDC biopsies analyzed with quantitative fluorescence microscopy. The average ratio of specific fluorescence in tumor versus normal tissue was 1.7 and 2.3 at 24 and 48 hours, respectively, after intravenous administration of porfimer.26 Porfimer enrichment in CC tissue should be adequate for PDT from day 1 to 4 after intravenous administration.
Wong Kee Song et al.7 showed a reduction of up to 60% of tumor volume after PDT with hematoporphyrin and with chlorine in nude mice inoculated with a human CC cell line. The PDT effect was evaluated in Buffalo female rats inoculated with rat hepatoma cells. The mean complete necrosis of a single session of PDT was 10.2 mm3.27 Neoadjuvant PDT of CC revealed that the tumoricidal depth of the PDT modality with porfimer was limited to 4- to 4.5-mm tissue penetration.28 Newer photosensitizers lead to deeper tumor necrosis and a shorter period of photosensitivity. Meso-tetrahydroxyphenyl chlorine is absorbed in the near-infrared spectrum (652 nm) and has the above characteristics.29
OUTCOME OF PDT FOR ADVANCED BDCS
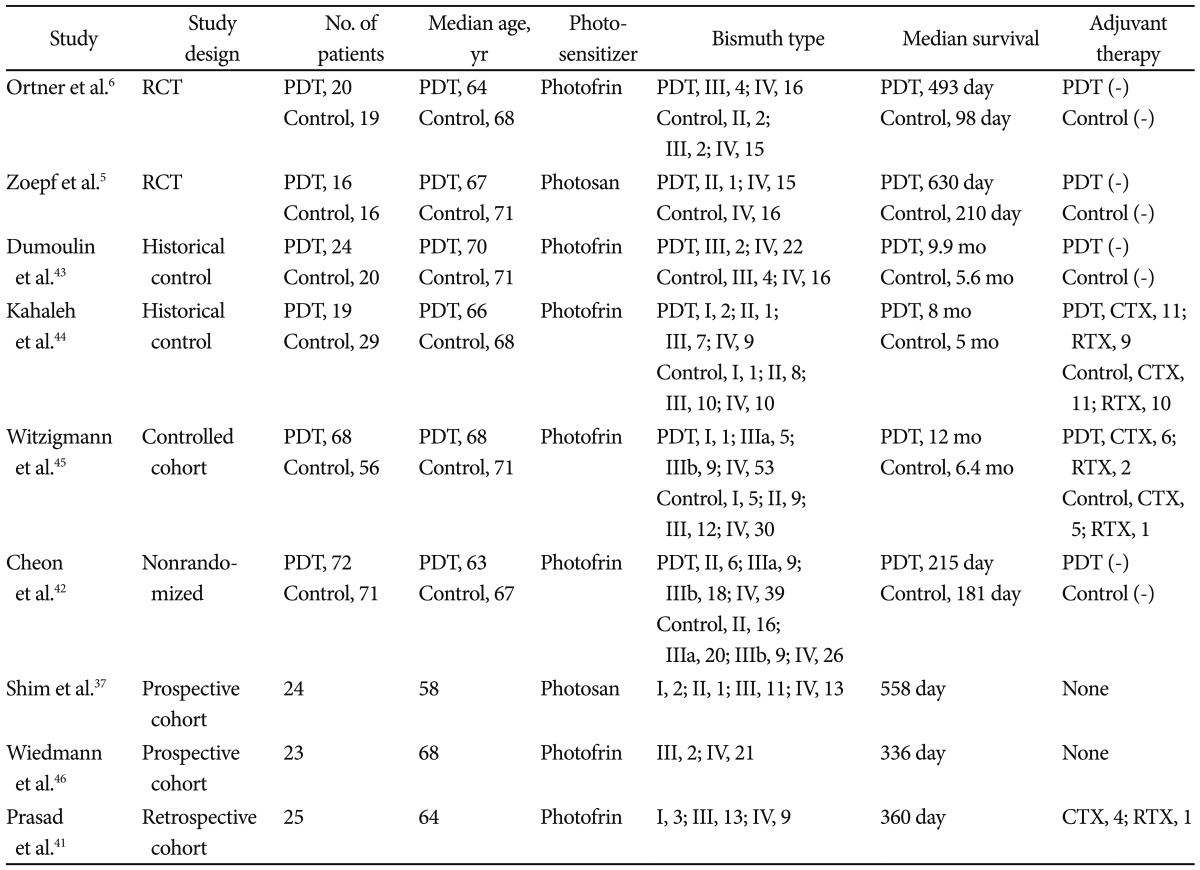
In numerous controlled and uncontrolled studies, the combination of PDT and biliary drainage has shown promising results in patients with unresectable hilar CC (Table 1).
The first case report of PDT for CC showed significant survival advantage over 4 years.30 In an early pilot study, Ortner et al.31 performed PDT in patients with unresectable CC Bismuth type III and IV who failed endoprostheses placement and had the poorest prognosis. Two days after intravenous application of a hematoporphyrin derivative Photofrin II, intraluminal photoactivation was performed cholangioscopically. Red light at 630 nm was emitted by an argon-dye laser and laser fibers of 400 ┬Ąm core diameter with flexible cylindrical diffuser tips of 2.5 and 4 lengths were used. With output of 800 mW, the light flux was 310 and 190 mW/cm2, and by changing irradiation time the resulting dose was 180 J/cm2. Bilirubin serum levels declined from 318┬▒72 to 103┬▒35 ┬Ąmol/L (p=0.0039) with no significant increase during the 2 monthly follow-ups. Quality of life indices improved dramatically (Karnofsky index 32.3%┬▒8.13% to 68.9%┬▒6.1%, p=0.0078; World Health Organization index 3.2┬▒0.36 to 1.7┬▒0.4, p=0.016; performance rating scale 13.6┬▒1.6 to 5┬▒0.93, p=0.0078) and remained stable during the follow-up. Thirty-day mortality was 0%, 1-year survival was 77.7%, and median survival time was 439 days.
In the prospective, open-label, randomized, multicenter study with a group sequential design comparing PDT in addition to stenting (group A) with stenting alone (group B) in patients with nonresectable CC was reported by Ortner et al.6 PDT resulted in prolongation of survival (group A, n=20, median 493 days; group B, n=19, median 98 days; p<0.0001). It also improved biliary drainage and quality of life. Authors concluded that PDT, given in addition to best supportive care, improves survival in patients with unresectable CC. The study was terminated prematurely because PDT was proved to be so superior to simple stenting treatment that further randomization was deemed unethical. Zoepf et al.5 conducted another prospective, randomized trial of 32 patients. The Photosan (SeeLab, Wesselburenerkoog, Germany) as a photosensitizer was administered. Median survival in PDT group was 630 days compared with 210 days for drainage alone. Compared with the study by Ortner et al.,6 the proportion of patients who received bilateral transpapillary stenting was low (10/18, 56%) and baseline patients' performance status was higher in Zoepf's study.32 Similar results were obtained by our previous study.7 Twenty patients who were treated with endoscopic biliary drainage alone (group A) and 27 patients treated with PDT under percutaneous cholangioscopy and additional percutaneous biliary drainage (group B) were analyzed retrospectively. The mean bilirubin level declined effectively in both group after treatment. One-year survival was 28% in group A, 52% in group B (p<0.05). Median survival time was 288 days in group A, 558 days in group B (p=0.0143).
One prospective study compared stenting with chemotherapy versus stenting with PDT for patients with hilar CC.33 The mean survival for the 17 patients treated with stenting and chemotherapy was 173 days, compared with 512 days for the 23 patients treated with stenting and PDT. As expected, 10 patients who were considered suitable for curative resection achieved longest survival of 1,278 days.
Recently, the effect of PDT on metal stent patency were analyzed in a retropsective study of 33 patients with unresectable CC by our group.34 We observed that the one session of PDT with metal stenting was associated with a significantly longer stent patency period (median 244┬▒66 and 177┬▒45 days, respectively, p=0.002) and patient survival (median 356┬▒213 and 230┬▒73 days, respectively, p=0.006) compared with the metal stent only group. The ability of PDT to destroy cancer cells and lessen cholestasis may prolong stent patency.
THE ROLE OF PDT FOR LOCAL RECURRENT TUMOR AFTER RESECTION OR AS NEOADJUVANT TREATMENT
We experienced a case whose survival time prolonged remarkably after the application of PDT for a postoperative recurrent tumor (Fig. 4). A small (n=8) uncontrolled study showed marked destruction of the recurrent tumor; 75% of patients were disease free after 2 years.35
Neoadjuvant PDT was evaluated in seven patients with advanced Bismuth type III and IV carcinoma, which were thought to be unresectable.28 After PDT, a curative resection could be performed in all patients; 83% were recurrence free after 1 year and 5-year survival was 71%. No relevant side effects of PDT occurred except for a minor intraoperative phototoxicity in one patient.
ASSESSMENT OF RESPONSE TO PDT
Evaluation of the therapeutic effects of PDT for patients with CC presents a number of challenges. Ortner et al.6 evaluated the therapeutic effect of PDT in cases of advanced BDC by comparing the tumor length before and after PDT using the 'mother-baby' cholangioscopic technique; however, it has been argued that their assessment was insufficiently objective. Although the authors23 reported reduced serum bilirubin levels after PDT, plastic biliary endoprostheses were inserted in all patients following the PDT procedure, making it difficult to assess the direct effect of PDT in reducing serum bilirubin. However, the PDT group might have mainly benefited from the number of ERCP sessions. For this reason, we do not consider the serum bilirubin level to be an objective parameter for assessing the response to PDT. Ductal CCs characteristically spread along or within the intrahepatic bile ducts, making it difficult to define the response to PDT based on changes in the tumor mass by computed tomography (CT). Similarly, it may be difficult to obtain objective results when the evaluation of the response to PDT is based solely on the extent of reopening of an occluded segment of the bile duct, as has been done in some studies.36 Previously, we assessed the thickness of the tumor mass before and after PDT treatment, measuring the thickest part of the tumor before the treatment and every month thereafter.37 The mean thickness of the bile duct masses, as measured by intraductal ultrasound, decreased from 8.7┬▒3.7 mm before PDT to 7.1┬▒2.0 mm at 1 month (p=0.176), to 7.1┬▒2.4 mm at 2 months (p=0.157), and declined significantly to 5.8┬▒2.0 mm at 3 months (p=0.046) after PDT.
In murine cancer models, some investigators have found that serum interleukin (IL)-6 levels correlate with tumor burden.38,39 Goydos et al.40 reported that serum IL-6 levels were correlated with the tumor burden as measured by CT in patients with CC; 2 weeks after tumor resection in three of 15 patients, IL-6 levels had dropped to undetectable levels in two patients and by almost tenfold in the third. Our results similarly demonstrated that IL-6 was significantly reduced (38.2┬▒9.9 pg/mL; p=0.008) 1 month after PDT compared with the pretreatment level (282.1┬▒121.8 pg/mL); tumor thickness also decreased following PDT. In contrast, IL-6 levels had not changed 1 month after ERBD.39
FACTORS ASSOCIATED WITH INCREASED SURVIVAL AFTER PDT
Recently, factors associated with patients' survival after PDT were analyzed in two studies.41,42 Prasad et al.41 conducted a retrospective study of 25 patients with unresectable CC treated with PDT in addition to biliary decompression. The presence of a visible mass on imaging studies, a lower serum albumin level and longer time between diagnosis and PDT predicted a poorer survival rate. Similar results were obtained in our retrospective analysis of 143 patients with hilar CC.42 We treated 72 patients with PDT and 71 patients with endoprotheses alone. We found that patients treated with PDT lived longer than those who were not treated with PDT (9.8 months vs. 7.3 months). Furthermore, lower pre-PDT bilirubin level, multiple PDT treatments and shortened time to treatment after diagnosis were significant predictors of improved survival. These two studies indicate the importance of early PDT after the diagnosis of unresectable CC.
CONCLUSIONS
Patients with unresectable CC had so far a very short life expectancy. PDT is the first palliative treatment option that has shown its efficacy in two randomized prospective studies. PDT improves survival, jaundice, and quality of life, is well tolerated and can be repeated without losing its efficacy.43-46 PDT seems to be a promising therapeutic approach for unresectable CC. It combines the aim to treat cholestasis and to reduce tumor growth. PDT, therefore, should be considered as a standard care for the palliation of CC. If the results are confirmed one could think of trying new photosensitizers with greater penetration depth and shorter photosensitivity, or using better drug targeting or combination therapies to induce more tumor necrosis. As PDT treatment is not available in all centers, patients should be referred to a specialized center with PDT availability. It is still not known whether radiotherapy and/or chemotherapy further improve the fate of PDT patients. It is now necessary to strengthen these data in an extended randomized multicenter study. PDT for recurrent tumors after surgery and neoadjuvant PDT is still experimental.