INTRODUCTION
Achalasia is an esophageal motility disorder affecting the smooth muscle layer of the esophagus and lower sphincter.1 It is characterized by incomplete lower esophageal sphincter (LES) relaxation, increased LES tone, and lack of peristalsis leading to difficulty in swallowing, regurgitation, and occasional chest pain.2 The cause of most achalasia cases is unknown.3
Treatment of achalasia focuses on forced relaxation of the LES by endoscopic or surgical methods. So far, endoscopic procedures including botulinum toxin injection and balloon dilation have been commonly performed as first-line therapies but are associated with limited efficacy and a high rate of recurrence.4 Although laparoscopic Heller myotomy with antireflux procedure is considered to be the standard treatment due to its excellent results and minimal invasiveness, dissection of the LES still requires several abdominal incisions and some sort of surgical antireflex procedure which are accompanied by a certain morbidity rate.5 Endoscopic myotomy of the LES was first reported by Ortega et al.6 in 1980. They used a modified needle-knife to incise the muscle from the luminal side. Despite excellent clinical results in 17 patients with achalasia, further examination of this method has not been pursued due to concerns about perforation or injury to surrounding structures which could not be reliably avoided. After almost three decades, Pasricha et al.7 described the feasibility of performing endoscopic myotomy through a submucosal tunnel in an animal model. Their novel method and excellent outcomes are expected to be useful for using this method to treat patients with achalasia. The first use of this procedure on humans was performed by Inoue et al.8 who coined the term "peroral endoscopic myotomy (POEM)" and reported favorable results from 17 achalasia patients. After that, Inoue and Kudo9 enrolled 43 patients and reported their results; other researchers also reported the results of POEM performed on humans.10-12 Most POEM procedures produced good clinical results without serious complications.8-12 However, there have been no reports of POEM performed in South Korea. Therefore, we studied the technical feasibility of performing POEM using a porcine model.
MATERIALS AND METHODS
Endoscopic procedure
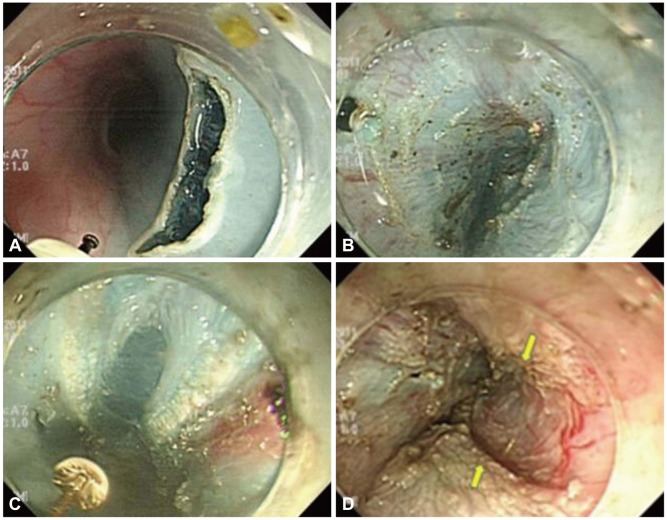
Two mini pigs (Micro-pig; Medi Kinetics, Pyeongtaek, Korea) ranging between 35 and 40 kg in weight were used for this study. Endoscopy was performed on the animals under general anesthesia. Normal saline (0.9%) was injected submucosally to raise the mucosa 10 cm above the gastroesophageal junction (GEJ) to 3 cm below GEJ and expand the submucosal space to facilitate safe mucosal incision. The initial mucosal incision was made 10 cm above the GEJ and a submucosal tunnel was subsequently created which extended to a point 3 cm below the GEJ. The tunnel was created primarily by using a dual knife (Olympus Medical Systems Co., Tokyo, Japan).
When we introduced the transparent cap-fitted endoscope into the submucosal space and applied some force, the endoscope tended to advance along the submucosal layer. The junction of the esophagus and stomach was readily recognized by identifying palisade vessels in the submucosal layer. Likewise, the configuration of the muscular fibers changed from one of uniform circularity to a more irregular arrangement. At this point, the circular muscle was cleanly incised in a distal to proximal fashion using several knives such as a dual knife, hook knife (Olympus Medical Systems Co.), and insulation-tipped (IT) knife (Olympus Medical Systems Co.) with direct visualization. The scope was then withdrawn into the lumen and the mucosal incision was closed using multiple resolution clips (Boston Scientific, Natick, MA, USA).
Postmyotomy period
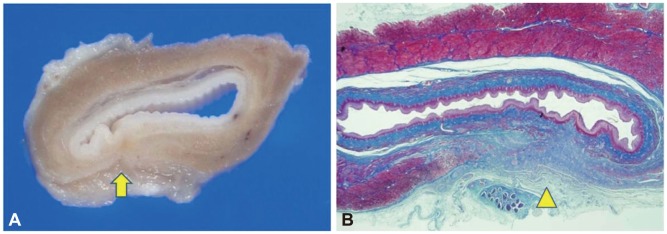
The two mini pigs were observed for 2 weeks after the procedure to assess their activity, feeding habits, vital signs, and body weight changes. During the 3 days after the procedure, amoxicilline and tetracycline were administered intramuscularly. Follow-up endoscopy was performed and the pigs were then euthanized with intravenous potassium chloride solution 2 weeks after the myotomy. Peritoneal, mediastinal, and thoracic examinations were performed postmortem to look for signs of peritonitis or infection. The esophagus was excised and fixed in formalin. Serial sections (2-mm thick) were then cut and processed to evaluate any histologic changes that occurred after the procedure. The slides were prepared using hematoxylin and eosin (H&E) stain as well as Masson's trichrome stain.
RESULTS
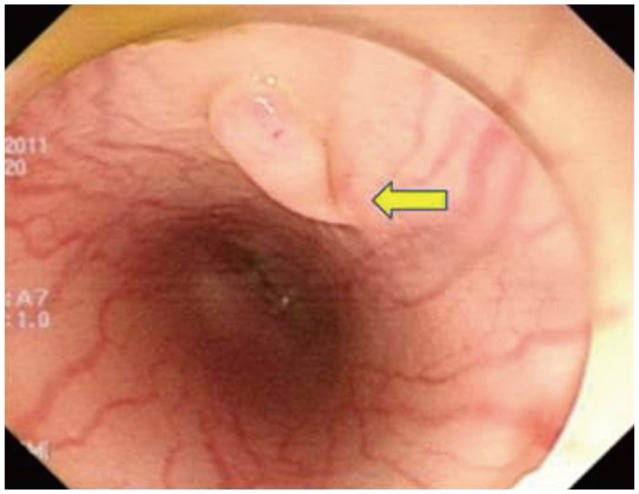
The procedure was successfully performed in the two mini pigs (Fig. 1). The total procedure time was approximately 100 minutes. Two weeks after the procedure, the animals appeared healthy with no evidence of fever or loss of appetite. Follow-up endoscopy revealed minor scarring at the mucosal entry site (Fig. 2). Necropsy revealed no evidence of mediastinitis or peritonitis. Grossly, there was minor retraction at the myotomy site. Histologic evaluation with Masson's trichrome stain showed that the inner circular muscle layer at the myotomy site was completely lost and replaced with fibrous tissue (Fig. 3).
DISCUSSION
Recently, the development of a submucosal tunnel method has made POEM technically feasible.13 POEM has already been performed on humans and produced excellent short-term results without serious complication.8-12 Both POEM and laparoscopic Heller myotomy target the LES for treating achalasia. Compared to laparoscopic myotomy, POEM has some advantages.14
First, POEM is an incisionless endoscopic treatment which is good for cosmetic reason. Second, myotomy length can be flexibly determined according to operator's choice. However, the upper mediastinum is difficult to approach using the standard laparoscopic technique. Third, POEM can be performed to selectively dissect the inner circular muscle fiber whereas full-thickness laparoscopic myotomy increase the possibility of vagal nerve or adjacent structure injury and reflux esophagitis.15 High LES pressure decreases to a normal range without any incision of the outer longitudinal muscle layer.8 Despite these advantages, POEM has several drawbacks.14 First, POEM is a newly developed technique; therefore, previous reports and clinical data on POEM safety and efficacy are lacking. To be considered an alternative to more invasive laparoscopic myotomy for treating achalasia, long-term POEM results are needed because true therapeutic success can only be judged on a long-term basis. Second, general anesthesia and complex preparations are needed to perform POEM. In addition, mean procedure time is similar to laparoscopic myotomy (approximately 2 hours). Therefore, the potential benefit of POEM might be marginal compared to laparoscopic myotomy. Third, antireflux technique could not be added to POEM, therefore theoretically it could induce more potential of esophagogastric reflux over laparoscopic myototomy with antireflux procedure. Fourth, achalasia is a very rare disease. In order to perform POEM successfully, sufficient practice is required and the learning curve is considerable. In our opinion, it is unreasonable to expect every gastroenterologist to perform POEM because of the substantial complication risk associated with the learning curve. Therefore, a referring system may be needed to maintain the quality of procedure performance.
According to a meta-analysis comparing endoscopic and surgical treatments for achalasia, surgical treatment improved symptoms and lowered LES pressure more than botulinum toxin injection or endoscopic balloon dilation.16 In terms of complications, the incidence of symptomatic perforation was 0.7% for surgical treatment compared to 1.6% for endoscopic balloon dilation.16 Therefore, laparoscopic management became the standard therapy for treating achalasia. In order for POEM to be regarded as the standard therapy for achalasia, high efficacy and a low complication rate should be demonstrated. Until now, POEM has been thought to be a promising treatment. According to recently published articles, the short-term symptom relief rate was 94% to 100%. During the procedure, pneumoperitoneum, and mediastinal exposure occurred; however, no patient developed serious complications.8-12
In spite of these recent studies, there is no report of POEM being performed in South Korea. Although current study evaluated POEM feasibility in an animal model, our results are meaningful because this was the first report in South Korea. Similar studies were carried out in many centers in South Korea but the outcomes have not been published into articles.
The esophagus of a mini pig looks very similar to that of a human. The wall of the porcine esophagus consists of four basic layer; mucosa, submucosa, muscularis propria, and serosa. Muscularis propria is composed of an inner circular and outer longitudinal smooth muscle layer.17 Some say that the pig esophagus is so fragile and muscularis propria is movable that it is not a good model to train esophageal endoscopic submucosal dissection.18 Nevertheless, porcine esophagus would be a good substitute for training POEM due to its anatomic similarity, and actually it has been widely used to study and train POEM.7,15,19
Based on our experience, we believe that several endoscopic techniques should be established to ensure POEM safety in humans. First, it is important to know which knife is appropriate for cutting the inner circular muscle. We used IT, hook, and dual knives in the present study; however, we felt that the IT and dual knives were associated with increased risk because we could not estimate the cutting depth properly. Inoue et al.8 used a triangle-tip knife which is considered relatively safe because it can selectively pull and cut the inner circular muscle. In addition, further studies about adequate myotomy length and depth of myotomy (full thickness versus partial thickness) are also required. Recently, the Korean Natural Orifice Transluminal Endoscopic Surgery Society held a hands on POEM training program which helped trainees to gain the skills needed to perform this procedure. POEM must be executed by adequately trained endoscopists. Before this technique can be performed by less experienced endoscopists, establishing strict protocols is mandatory.
Besides endoscopic techniques, we should also account for various factors such as ventilator and back-up systems. We believe that general anesthesia with positive pressure ventilation and CO2 insufflation through the endoscope is mandatory for performing POEM due to the high possibility of mediastinal emphysema development during the procedure.8 Under conscious sedation with room air, mediastinal exposure induces pneumomediastinum and mediastinal emphysema which may endanger the patient. In addition, cooperation with thoracic surgeons is mandatory when complications arise during the procedure.
Our study has several limitations. First, we used only two nonachalasic pigs. Pig model may not be appropriate and not be equivalent to achalasia patients. Therefore, it is thus difficult to comprehensively verify the safety and feasibility of POEM. Second, we did not measure esophageal manometry before and after the procedure. Therefore, we could not evaluate POEM efficacy. However, sharing our experiences will give trainee endoscopists useful information which obviate unwanted complications and shorten learning curve.
In conclusion, our study demonstrated that POEM is a technically feasible procedure. Until now, excellent POEM short-term outcomes without serious complications have been reported in many publications. However, before POEM can be safely performed on human patients, further studies about technical methods and appropriate instrumentation are required along with long-term follow-up data.