The spleen is a lymphopoietic gland that plays an essential role in the reticuloendothial system. Although uncommon, the spleen can be the site of infectious and malignant diseases, such as lymphoma [1]. While cross-sectional imaging can help suggest a possible diagnosis, histological and immunochemical evaluation is generally necessary for definitive diagnosis [2].
Strategies to obtain splenic mass samples include surgery and percutaneous and endoscopic approaches [3-5]. Percutaneous ultrasound-guided biopsy is the standard technique for intra-abdominal organs; however, in experienced hands, endoscopic ultrasound (EUS)-guided biopsy can also be a safe and effective technique for acquiring tissue samples from intra-abdominal organs [6]. Utilizing proximity to the gastrointestinal tract, EUS provides a novel approach to sampling a splenic mass as the core biopsy needle traverses less tissue, and may reduce the risk of hemorrhage and hematoma [5]. Previously, evaluation using EUS-guided biopsy has been limited to cytodiagnosis with fine needle aspiration; however, the use of 19 G core biopsy needles maintains tissue integrity, allowing for immunochemical and histological assessment [7].
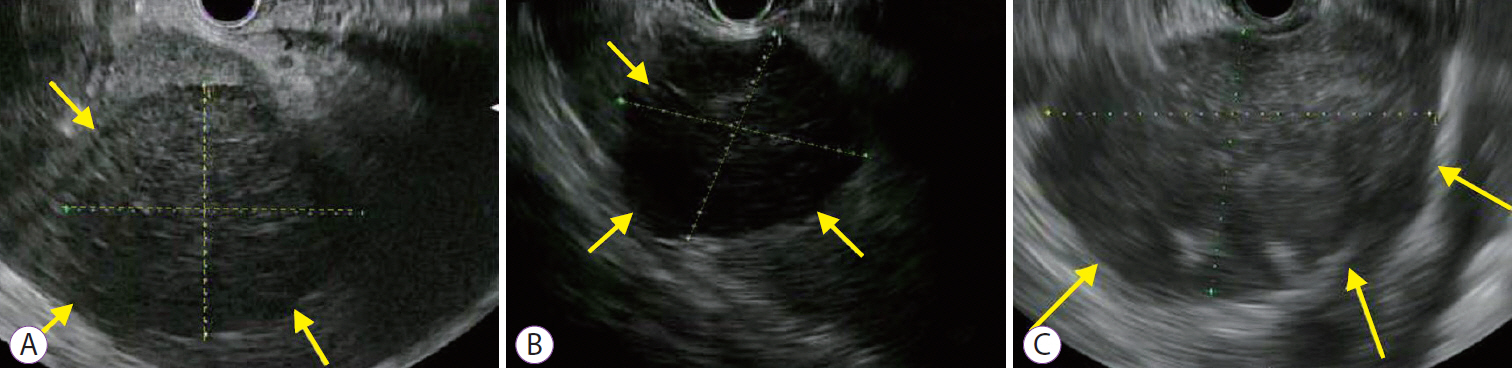
Three patients underwent EUS-guided spleen biopsy (SB) for suspected lymphoma. The median age (range) of the 3 patients was 64 (37ŌĆō77) years. EUS was performed using a standard linear-array echoendoscope (GF-UE180-AL5; Olympus America, Center Valley, PA, USA). EUS imaging is shown for all 3 patients in the Fig. 1. After identifying the spleen with EUS, a 19 G needle (AcquireŌäó 19 G [Boston Scientific Corporation, Santa Clarita, CA, USA], for case 1, and SharkcoreŌäó 19 G [Medtronic, Dublin, Ireland] for cases 2 and 3) was used to obtain core tissue from the splenic mass. One pass with 2 actuations was performed under wet suction. After extraction, core tissue samples were fixed with formalin and sent for flow cytometry. We recorded the total core tissue length, longest intact specimen length, number of cores, and histologic diagnosis.
The median (range) length along the greatest sample dimension was 4.5 cm (3.9ŌĆō7.1 cm). The median (range) number of cores obtained was 8 (8ŌĆō11). The median (range) total specimen length and longest specimen length was 8.5 cm (8ŌĆō8.5 cm) and 2.5 cm (2ŌĆō2.5 cm), respectively. Immunochemical staining showed: CD 10+ and BCL-6+; CD 10ŌĆō and BCL-2+; and CD10 +, CD20+, Ki67+, IgH:MYCt (8:14) respectively, in the 3 patients. A definitive diagnosis of lymphoma was made in all patients and there were no complications using EUS-guided SB.
EUS has been described in the assessment of splenic masses but utilization consisted of fine-needle aspiration [5,8]. Use of core biopsy needles for EUS-guided SB allows for acquisition of larger samples, and enables histological analysis and immunochemical staining [7]. By using a 19 G rather than a 22 G needle, our novel approach is associated with fewer passes and decreased risk bleeding while maximizing tissue quality [9]. Using a core needle, biopsies of the spleen have a sensitivity between 86-91% and a specificity between 91-97% [4,9].
In comparing the complications of techniques used for evaluation of splenic masses, surgery is associated with the highest morbidity rates, probably because of infection [10]. At 8.2%, the estimated overall complication rate is lower in image-guided percutaneous spleen biopsy [4]. With use of smaller needles for image-guided percutaneous spleen biopsy, the complication rate decreases to 2.4% [9]. EUS-guided SB allows for not only direct evaluation of splenic masses, but also surrounding organs. The results of our study showed that EUS-guided SB in experienced hands can be safe and effective in the evaluation of splenic masses.
EUS-guided core biopsy is an effective diagnostic approach in patients with splenic masses. Additional studies are needed to assess the safety of EUS-SB.