INTRODUCTION
Ischemic colitis is an inflammatory disease caused by the lack of colonic blood supply.1,2 Being one of the most common form of intestinal ischemias, ischemic colitis is manifested with unspecific clinical symptoms such as acute abdominal pain, diarrhea, or hematochezia. The major causes include hypotension due to dehydration or bleeding and decreased blood flow.3 Ischemic colitis occurs mostly in the elderly because of age-related tortuosity of the colonic arteries and so on.4 Factors other than age include diseases of decreased blood flow; small vessel etiology due to underlying diseases such as thrombosis, hypertension, and diabetes mellitus, iatrogenic causes such as surgery, or drugs.2,3 Drug, especially estrogen, has been reported as the cause of ischemic colitis in Western countries.3,5,6 But in Korea, Estrogen is a very rare cause of ischemic colitis. We report a case of ischemic colitis with a review of literature, developing after taking combined oral contraceptives without particular comorbidity in a 26-year-old American female living in South Korea.
CASE REPORT
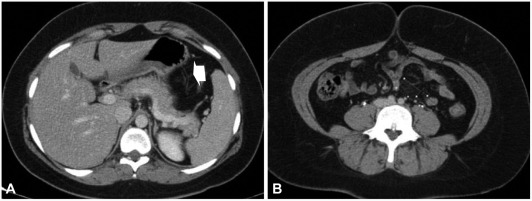
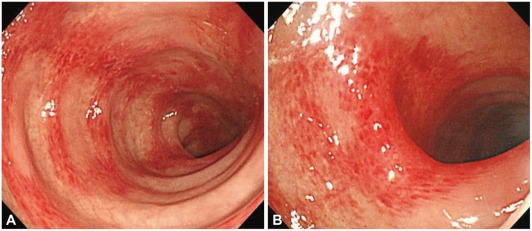
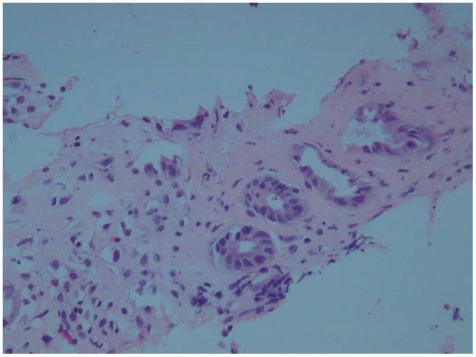
A 26-year-old foreign female visited the hospital for sudden occurrence of lower abdominal pain during sleep with more than 10 times of diarrhea, and 3 times of hematochezia. There was no specific finding from her past medical and social history. She is unmarried and has been working as an elementary school English teacher in South Korea for 2 years with generally good health. Blood pressure 130/90 mm Hg, pulse rate 60/min, respiratory rate 20/min, body temperature 37.3Ōäā, height 173 cm, weight 99 kg, and body mass index (BMI) 33 were measured at admission. Physical examination revealed acute ill looking appearance, with soft abdomen but tenderness on the lower abdomen. Complete blood count test showed leukocyte 8,630/mm3, hemoglobin 12.8/dL, and platelet count 210,000/mm3. Blood chemistry was analyzed as total protein 7.2 g/dL, albumin 4.6 g/dL, total bilirubin 0.3 mg/dL, AST 17 U/L, ALT 17 U/L, alkaline phosphatase 47 U/L, amylase 140 U/L, lipase 43 U/L, blood urea nitrogen 10.9 mg/dL, creatinine 0.9 mg/dL, ESR 10 mm/hr, and CRP 0.77 mg/dL. Blood coagulation test revealed PT/aPTT of 10.7/24.2 seconds. Stool examination showed WBC 0-2/HPF and RBC 0-2/HPF, with negative culture result. Urine Pregnancy test was also negative. Abdominal computerized tomography taken on the day of admission showed small amount of ascites in the pelvic cavity and blood vessels were normal without unusual finding (Fig. 1). Sigmoidoscopy on day 2 of admission revealed normal rectum and sigmoid colon, but erythemas and petechias were found along the longitudinal axis from the descending colon to the splenic flexure. Transverse colon appeared normal (Fig. 2). Biopsy was performed at the descending colon, which revealed loss of the surface epithelium, mucosal necrosis, and hyalinization of lamina propria compatible with ischemic colitis (Fig. 3). The patient was managed conservatively with bowel rest, intravenous fluids, and empirical antibiotics. Symptoms of abdominal pain, diarrhea, and hematochezia were relieved and the patient started soft diet on day 4 of admission, and she was discharged on day 5 of admission. After 2 months of asymptomatic period, she visited the outpatient department again with the recurrence of abdominal pain and hematochezia. On repeated carefyl history taking, she gave drug history about taking combined oral contraceptives (mercilon; combined ethinyl estradiol and desogestrel) since coming to Korea. Also she remembered intermittent abdominal pain without reasonable causes before the last hospitalization. The new onset abdominal pain recurred 2 days ago with hematochezia after restarting the use of oral contraceptives 1 month ago. We advised her to stop the use of oral contraceptives after explaining that it might be the possible cause of ischemic colitis. Symptoms improved after conservative care for a few days. She stopped using oral contraceptives thereafter. We confirmed by a phone call 6 months later that she was faring well without any recurrence of abdominal pain or hematochezia.
DISCUSSION
Ischemic colitis is caused by various predisposing factors including old age, hypotension and decreased blood flow due to diseases, cardiogenic thromboembolism, myocardial infarction, major cardiovascular surgery, hypercoagulable state, drugs, long-distance running, constipation, vasospasm, and snake venom.2,3 The patient of this study was hard to find a reasonable cause of ischemic colitis because she did not have any risk factor and history taking was not sufficient enough to find the combined oral contraceptives taking in unmarried person. Recurrent symptom onset made it possible to diagnose oral contraceptive related ischemic colitis. It could be said, therefore, that finding predisposing factors of ischemic colitis is the most important to prevent further recurrence. It is impossible and sometimes not necessary to inspect all the above mentioned causes for every ischemic colitis patients. Ischemic colitis in the elderly is rarely caused by hypercoagulable state, so the test of it is generally not recommended.3 Younger patients, however, require tests on hypercoagulable state and detailed history taking for drugs.
Venous thrombosis caused by estrogen in oral contraceptives was first reported in the early 1960s.7 In 1990s, it was reported that women with genetic defects such as factor V Leiden mutation, prothrombin 20210A, and deficiency of anti-thrombin, protein C and protein S was more susceptible to venous thrombosis while using oral contraceptives.8-10 Oral contraceptives change the blood concentration of lots of proteins involved in the coagulation.11 It is not clear how these changes of coagulation-related factors clinically affect the venous thrombosis.12 Based on the available studies, the mechanism of venous thrombosis caused by oral contraceptives could be explained by the activated protein C (APC) resistance.12 The APC resistance is related to the blood concentration of protein S, a tissue factor pathway inhibitor, and has a positive relation with sex hormone binding globulin concentration.12-14 Each oral contraceptives causes venous thrombosis at different frequencies. It was reported that the risk of venous thrombosis depends on the dose of estrogen before 1995.12 It was reported later that progesterone involves in the development of venous thrombosis, leading to a new concept of 'total estrogenicity' combining the antiestrogenic effects of estrogen and progesterone.15 If the estrogen is fixed on a certain dose, the frequency of venous thrombosis depends on the combined progesterone, so that the 3rd (gestodene or desogestrel) and 4th (cyproterone acetate or drospirenone) generation of progesterone increase the risk of venous thrombosis 1.5-4 times higher than the 2nd generation progesterone (levonorgetrel).12 Moreover, obese females have 2.4-times higher risk of thrombosis than females with the normal weight. Obese females taking oral contraceptives have the risk of developing thrombosis 24 times higher than normal weight females not taking oral contraceptives.16 Our patient was taking combined oral contraceptives including desogestrel, which causes venous thrombosis more often, and was obese with BMI value of 33, contributing the increased risk of thrombosis than normal weight female who were taking the same contraceptives.
Patients manifest rapid onset of abdominal pain, tenderness around the affected bowel area, and mild to moderate hematochezia, as in this case.3 Any part of the colon could be involved, but around 75% shows left colon lesion, 25% of them including splenic flexure.17 Our case also showed typical descending colon lesion including splenic flexure. The diagnosis of ischemic colitis is initially suspected by typical clinical presentation, but the clinical manifestations are similar with other various diseases, making it hard to diagnose only by clinical suspicion. Computerized tomography is useful to exclude other diseases presenting similar symptoms but difficult to make a definite diagnosis. Colonoscopy, on the other hand, is more sensitive by inspecting the location, and mucosal changes and getting biopsy, which are important in the diagnosis of ischemic colitis.3 Our patient was also unable to be diagnosed as ischemic colitis by the abdominal computerized tomography but sigmoidoscopy enabled the definite diagnosis.
Single linear ulcer along the longitudinal axis is a distinctive colonoscopic finding suggestive of ischemic colitis, along with other findings of pale, edematous, petechial mucosa to hemorrhagic nodule and gangrene appearing in various degrees. The histologic patterns of ischemic colitis include mucosal coagulation necrosis and hemorrhage, neutrophil infiltration, and granulation tissue formation; these patterns are not specific for diagnosis but helpful for predicting the disease progression.18,19 This patient showed mucosal necrosis and hyalinization of lamina propria. Angiography is only helpful for the diagnosis of ischemic colitis caused by thrombosis. Accordingly, ischemic colitis should be diagnosed in combination of clinical manifestations, radiologic images, and colonoscopic findings.3
Treatment of ischemic colitis depends on the clinical manifestations and colonoscopic findings. Most of patients with ischemic colitis improve with conservative managements such as bowel rest, intravenous fluids, and empirical antibiotics. Some cases may require surgery for gangrenous colitis or stricture.20 Prognosis depends on the extent of injury and comorbidities, but around 85% of patients are improved within 1-2 days of conservative management and fully recovered within 2 weeks. About 20% of patients progress to a peritonitis requiring a surgery. Our patient recovered with conservative management and did not have the recurrence of the disease after quitting the possible cause. This case represents the repetitive ischemic colitis due to the use of oral contraceptives in young healthy female without comorbidity, suggesting the importance of precise history taking including oral contraceptives and other risk factors for thrombosis.