AbstractEndoscopic submucosal dissection (ESD) was developed for the en bloc resection of large early gastrointestinal neoplasms. A disadvantage of ESD is its technical difficulty, which requires advanced skills and is associated with a higher rate of complications. Endoscopic variceal obturation (EVO) using cyanoacrylate has emerged as the initial treatment of choice for acute gastric variceal bleeding. This procedure achieves hemostasis in 90% of cases. A 52-year-old patient with Child A alcoholic liver cirrhosis presented with early gastric cancer in the cardia and type 1 isolated gastric varices in the fundus. The two lesions were so close together that treatment was not easy. The lesions were managed successfully with a combination of ESD and EVO using cyanoacrylate.
INTRODUCTIONEndoscopic submucosal dissection (ESD) is an effective procedure that allows the en bloc resection of premalignant lesions and early gastric cancer (EGC) in the stomach.1-3 However, ESD is technically difficult and time consuming.4,5 Major complications of ESD are bleeding and perforation and its complication rate is higher than that of conventional endoscopic mucosal resection.6-8 Gastric variceal bleeding is associated with more massive bleeding than esophageal variceal bleeding.9 A fundal varix is closely related to an increased risk of bleeding.9 Endoscopic variceal obturation (EVO) using N-butyl-2-cyanoacrylate (Histoacryl) is effective for the treatment of gastric varices bleeding.10-12 This is, to our knowledge, the first report of the successful treatment of EGC adjacent to a fundal varix by ESD and EVO using cyanoacrylate.
CASE REPORTA 52-year-old male was referred to our tertiary center for the treatment of EGC. He had a history of alcoholic liver cirrhosis. His liver function was evaluated as Child A. Laboratory evaluation revealed the following: AST/ALT, 30/30 IU/L; albumin, 4.2 g/dL; total bilirubin, 0.7 mg/dL; prothrombin time (INR), 11.9 seconds (1.03); white blood cell count, 6,100/┬ĄL; hemoglobin, 12.7 g/dL; and platelet count, 152,000/┬ĄL. Upper endoscopy revealed a depressed lesion and an isolated gastric varix in the vicinity of the cardia (Fig. 1). There were no esophageal varices. Forceps biopsy on the depressed lesion showed differentiated adenocarcinoma and endosonography suggested that the depth of tumor invasion was superficial to the submucosal layer around the varices.
In this case, the surgical option was a total gastrectomy, due to the location of the lesion. However, the patient decided against surgical treatment and ESD was chosen. Treatment priority was given to the fundal varix to prevent bleeding during the ESD (Fig. 2). Endoscopic therapy with tissue adhesive was used. Typically, 2 mL of a 1:1 mixture of cyanoacrylate (B. Braun, Tuttlingen, Germany) and Lipiodol (Guerbet, Aulnay-sous-Bois, France) was administered. After 2 weeks, Doppler endosonography showed absence of blood flow in the varix. Then, ESD was performed with a forward-viewing endoscope (EG 2990i; Pentax, Tokyo, Japan) (Fig. 3). Prophylactic blood transfusion of platelets or fresh frozen plasma was not performed before ESD. After making several marking dots outside the lesion with a dual knife (KD-650Q; Olympus, Tokyo, Japan), ESD was performed with the dual knife and an insulated-tip diathermic knife (KD-611L; Olympus). When a circumferential incision was made around the varices, no bleeding was seen. The submucosal dissection was performed in forced coagulation mode (VIO 300D; ERBE, T├╝bingen, Germany). Precoagulation of the submucosal vessels with a Coagrasper (FD-410LR; Olympus) was used to prevent severe bleeding. The en bloc resection was accomplished with a total procedure time of 90 minutes. Histology revealed a moderately differentiated adenocarcinoma measuring 24├Ś12 mm size with disease-free margins (Fig. 4A). The depth of submucosal invasion was 150 ┬Ąm, with no lymphovascular invasion (Fig. 4B). Follow-up endoscopy demonstrated a healing ulcer with no evidence of recurrence 2 months after the ESD (Fig. 5).
DISCUSSIONGastric varices are classified as gastroesophageal or isolated gastric varices.9 Type 1 gastroesophageal varices, which extend to the lesser curvature, are the most common. Type 2 gastroesophageal varices, which extend to the greater curvature, are associated with high mortality. Type 1 isolated gastric varices, which involve only fundal varices, have a high incidence of bleeding. Type 2 isolated gastric varices are gastric varices that arise at sites other than the fundus. As a type 1 isolated gastric varix was detected in our case, it was essential to manage the varix before performing ESD. EVO using cyanoacrylate is superior to other modalities, such as endoscopic injection sclerotherapy,13 endoscopic variceal ligation,14 and transjugular intrahepatic portosystemic shunt15 in the management of bleeding gastric varices. Cyanoacrylate abruptly forms a hard substance when exposed to moisture, such as blood. Thus, after its injection, cyanoacrylate occludes the lumen, resulting in rapid hemostasis and prevention of recurrent bleeding.16-18
During ESD, acute bleeding occurs more frequently in the upper stomach.19,20 According to a recent multicenter study,2 the en bloc resection rate of EGC was 88.6% and the mean ESD time was 64.8 minutes in the upper stomach, compared with 96% and 44.1 minutes in the lower stomach. In this case, the EGC was located in the cardia. Thus, it was difficult to keep the knife blade parallel to the muscle layer to prevent perforation. Bleeding was controlled with the routine coagulation of visible vessels. When ESD is applied to patients with liver cirrhosis, postoperative bleeding and long operating time are of concern, which may be due to the increased bleeding risk associated with a low platelet count or prolonged prothrombin time.21 However, a recent study by Kwon et al.22 showed that ESD for the treatment of early gastric neoplasia may be as effective and tolerable in patients with liver cirrhosis as those in patients without.
References1. Ono H. Early gastric cancer: diagnosis, pathology, treatment techniques and treatment outcomes. Eur J Gastroenterol Hepatol 2006;18:863ŌĆō866. 16825902.
2. Chung IK, Lee JH, Lee SH, et al. Therapeutic outcomes in 1,000 cases of endoscopic submucosal dissection for early gastric neoplasms: Korean ESD Study Group multicenter study. Gastrointest Endosc 2009;69:1228ŌĆō1235. 19249769.
3. Isomoto H, Shikuwa S, Yamaguchi N, et al. Endoscopic submucosal dissection for early gastric cancer: a large-scale feasibility study. Gut 2009;58:331ŌĆō336. 19001058.
4. Kakushima N, Fujishiro M, Kodashima S, Muraki Y, Tateishi A, Omata M. A learning curve for endoscopic submucosal dissection of gastric epithelial neoplasms. Endoscopy 2006;38:991ŌĆō995. 17058163.
5. Yamamoto S, Uedo N, Ishihara R, et al. Endoscopic submucosal dissection for early gastric cancer performed by supervised residents: assessment of feasibility and learning curve. Endoscopy 2009;41:923ŌĆō928. 19802773.
6. Messmann H, Probst A. Management of endoscopic submucosal dissection complications. Endoscopy 2009;41:712ŌĆō714. 19670140.
7. Probst A, Golger D, Arnholdt H, Messmann H. Endoscopic submucosal dissection of early cancers, flat adenomas, and submucosal tumors in the gastrointestinal tract. Clin Gastroenterol Hepatol 2009;7:149ŌĆō155. 19032991.
8. Oka S, Tanaka S, Kaneko I, et al. Advantage of endoscopic submucosal dissection compared with EMR for early gastric cancer. Gastrointest Endosc 2006;64:877ŌĆō883. 17140890.
9. Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology 1992;16:1343ŌĆō1349. 1446890.
10. Huang YH, Yeh HZ, Chen GH, et al. Endoscopic treatment of bleeding gastric varices by N-butyl-2-cyanoacrylate (Histoacryl) injection: long-term efficacy and safety. Gastrointest Endosc 2000;52:160ŌĆō167. 10922085.
11. Akahoshi T, Hashizume M, Shimabukuro R, et al. Long-term results of endoscopic Histoacryl injection sclerotherapy for gastric variceal bleeding: a 10-year experience. Surgery 2002;131(1 Suppl):S176ŌĆōS181. 11821807.
12. Dhiman RK, Chawla Y, Taneja S, Biswas R, Sharma TR, Dilawari JB. Endoscopic sclerotherapy of gastric variceal bleeding with N-butyl-2-cyanoacrylate. J Clin Gastroenterol 2002;35:222ŌĆō227. 12192197.
13. Sarin SK, Jain AK, Jain M, Gupta R. A randomized controlled trial of cyanoacrylate versus alcohol injection in patients with isolated fundic varices. Am J Gastroenterol 2002;97:1010ŌĆō1015. 12003381.
14. Lo GH, Lai KH, Cheng JS, Chen MH, Chiang HT. A prospective, randomized trial of butyl cyanoacrylate injection versus band ligation in the management of bleeding gastric varices. Hepatology 2001;33:1060ŌĆō1064. 11343232.
15. Procaccini NJ, Al-Osaimi AM, Northup P, Argo C, Caldwell SH. Endoscopic cyanoacrylate versus transjugular intrahepatic portosystemic shunt for gastric variceal bleeding: a single-center U.S. analysis. Gastrointest Endosc 2009;70:881ŌĆō887. 19559425.
17. Tripathi D, Ferguson JW, Therapondos G, Plevris JN, Hayes PC. Review article: recent advances in the management of bleeding gastric varices. Aliment Pharmacol Ther 2006;24:1ŌĆō17. 16803599.
18. Ryan BM, Stockbrugger RW, Ryan JM. A pathophysiologic, gastroenterologic, and radiologic approach to the management of gastric varices. Gastroenterology 2004;126:1175ŌĆō1189. 15057756.
19. Okano A, Hajiro K, Takakuwa H, Nishio A, Matsushita M. Predictors of bleeding after endoscopic mucosal resection of gastric tumors. Gastrointest Endosc 2003;57:687ŌĆō690. 12709698.
20. Jeon SW, Jung MK, Cho CM, et al. Predictors of immediate bleeding during endoscopic submucosal dissection in gastric lesions. Surg Endosc 2009;23:1974ŌĆō1979. 18553202.
21. Ogura K, Okamoto M, Sugimoto T, et al. Efficacy and safety of endoscopic submucosal dissection for gastric cancer in patients with liver cirrhosis. Endoscopy 2008;40:443ŌĆō445. 18494135.
22. Kwon YL, Kim ES, Lee KI, et al. Endoscopic treatments of gastric mucosal lesions are not riskier in patients with chronic renal failure or liver cirrhosis. Surg Endosc 2011;25:1994ŌĆō1999. 21136091.
Fig.┬Ā1(A) Endoscopic views of the isolated gastric varix. (B) Early gastric cancer adjacent to the varix. 
Fig.┬Ā2(A) Endoscopic cyanoacrylate therapy of the isolated gastric varix was performed. The picture shows cyanoacrylate with lipiodol filling the varix in fluoroscopic view. (B) On endosonography 2 weeks later, there was no blood flow in the varix. 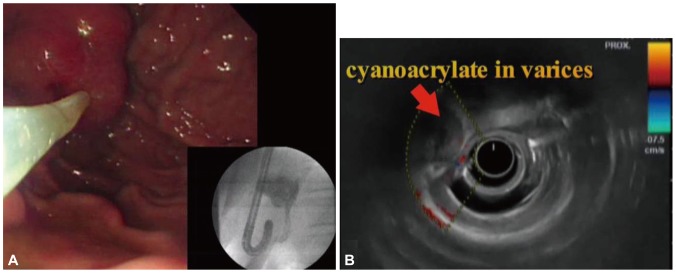
Fig.┬Ā3Endoscopic submucosal dissection of the early gastric cancer (EGC) showing the EGC and treated varix before (A) endoscopic submucosal dissection, (B) the incision with muscle exposure, (C) the incision above the varix, and (D) a large artificial ulcer after the resection. 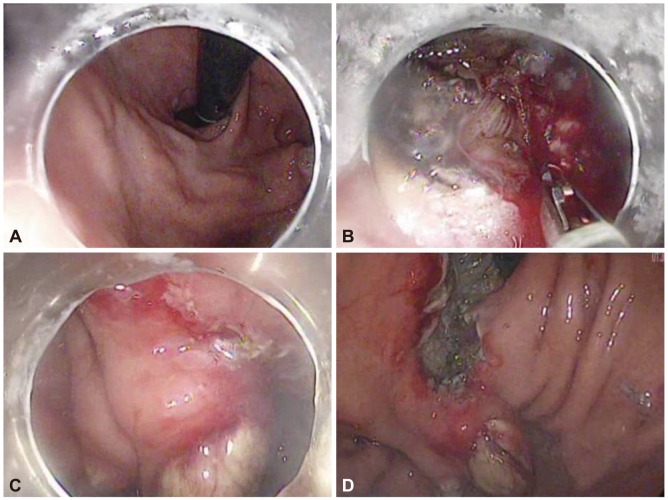
Fig.┬Ā4(A) Pathologic mapping of the specimen shows the tumor in the center of the specimen. The tumor was completely resected with free lateral and vertical margins. The orange area depicts the area where the tumor invaded no further than the mucosal layer and the purple area show the area of submucosal invasion. (B) Microscopic findings revealed adenocarcinoma with minimal submcosal invasion, to a depth of 150 ┬Ąm (H&E stain, ├Ś100). 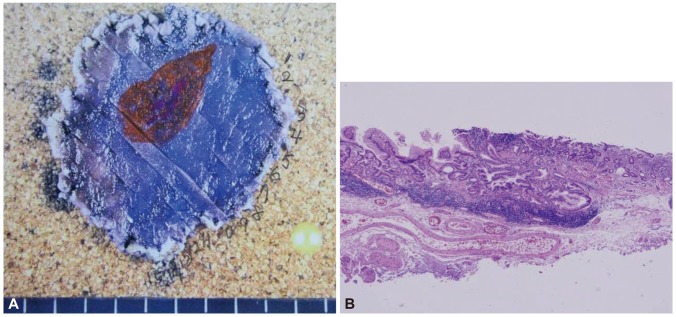
|
|
||||||||||||||||||||||||||||||||||||
