INTRODUCTION
Extra-nodal marginal zone B-cell lymphoma (mucosa-associated lymphoid tissue, MALT lymphoma) belongs to indolent lymphomas and has good prognosis. Approximately 70% to 90% of gastric MALT lymphomas are associated with Helicobacter pylori infection.1 And H. pylori eradication can induce complete regressions of gastric MALT lymphomas in about 60% to 95% of patients.2,3 Some reports suggested that low-grade MALT lymphomas can transformed to high-grade diffuse large B-cell lymphomas (DLBCLs), which is the important cause of death in patients with gastric MALT lymphomas.4,5 We report two cases of DLBCLs in the cervical lymph nodes that occurred 10 years and 1.5 years after diagnosing low-grade gastric MALT lymphomas.
CASE REPORTS
Patient 1
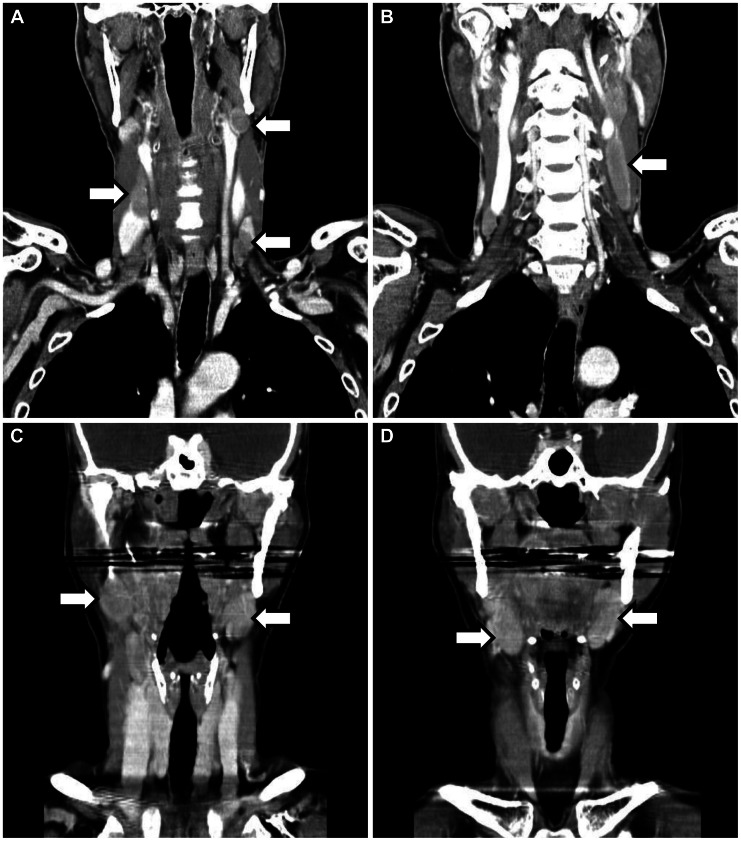
In June 1997, a 52-year-old man without any medical history was referred to the department of gastroenterology because of gastric subepithelial tumor. Esophagogastroduodenoscopy (EGD) demonstrated 1 cm sized subepithelial tumor on the proximal antrum anterial wall (Fig. 1A). Biopsies showed chronic gastritis only. He was admitted and diagnostic endoscopic mucosal resection was done (Fig. 1B). The pathologic finding was diffuse aggregation of monomorphic lymphoid cells without any large cell or small cell component in the submucosal layer (Fig. 2A, B). Immunohistochemical staining showed atypical lymphoid cells that were positive for pan B and lamda, but negative for pan T, bcl-2 and kappa. Ki-67 labeling index was 5%. These findings suggested his diagnosis was low-grade MALT lymphoma. Abdomen and pelvic computed tomography (CT), chest CT, bilateral bone marrow examination and F-18-fluorodeoxyglucose positron emission tomography (PET) were done and there were no involvement of disease. Although rapid urease test (RUT), serologic test for H. pylori, urea breath test (UBT) and H. pylori culture of gastric mucosa were all negative, he took triple regimen of H. pylori eradication including clarythromycin, amoxicillin, and proton-pump inhibitor (PPI) for 14 days. After the H. pylori eradication was completed, subtotal gastrectomy was done. In the pathologic review of surgical specimen, there were no residual tumor cells. We performed esophagogastroscopy, abdomen and pelvic CT and PET every 6 months for 2 years and then we performed the same examination every 12 months for 4 years. He had been followed without evidence of disease recurrence for 6 years, after which he stopped visiting our hospital. In May 2007, he was referred to our hospital again complaining of throat pain and neck mass. Neck CT showed multiple enlarged lymph nodes in bilateral cervical nodes (Fig. 3A, B). The largest diameter of the involved lymph node was 6 cm. Excisional biopsy showed diffuse infiltration of large atypical lymphoid cells (Fig. 2C, D), which were positive for CD 20, but not for CD 3, CD10, CD 56, ALK-1, or CD 21. Ki-67 labeling index was 90%. These findings suggested DLBCL. EGD and full staging evaluation showed no involvement of other site. He was diagnosed with stage IIA DLBCL and received three cycles of systemic chemotherapy including rituximab, cyclophospamide, doxorubicin, and vincristine (R-CHOP), followed by 3,600 cGy radiotherapy. He achieved and maintained complete remission until recent days.
Patient 2
In June 2005, a 55-year-old woman with hypothyroidism was referred to our department for epigastric discomfort from the department of endocrinology. EGD demonstrated mucosal surface granularity and 1 cm size mucosal elevation which had central depression on the lower body anterior wall (Fig. 1C). Biopsy showed diffuse infiltration of atypical lymphocyte aggregation and glandular invasion of lymphocytes. Immunohistochemical staining showed atypical lymphoid cells that were positive for CD 20 but negative for CD 3, Ki-67 labeling index was 8%. Abdomen CT, chest CT, bilateral bone marrow examination, and PET were done for staging and there were no disease involvement at any other site. Pathologic diagnosis was low grade MALT lymphoma of the stomach at stage IE. RUT and UBT were positive. The patient took H. pylori eradication including clarythromycin, amoxicillin, and PPI for 14 days. Six months later, EGD was followed and confirmed complete histologic remission as well as endoscopic remission. RUT and UBT were all negative. In August 2006, 12 months after the first visit, EGD showed multiple erosions on the proximal antrum (Fig. 1D). Biopsy showed recurred MALT lymphoma and H. pylori was also seen. RUT was positive, too. She took the second eradication of H. pylori infection with the same regimen during the same period of time. In November 2006, she visited our hospital complaining of palpable mass on both the lateral neck. Neck CT showed bilateral cervical lymph node enlargement (Fig. 3C, D). Excisional biopsy showed diffuse infiltration of large atypical lymphoid cells. Immunohistochemical staining was positive for CD 20 and CD 79 but negative for CD 3. Ki-67 labeling index was 80%. These findings suggested DLBCL. In the staging work-up, there was no disease involvement except for both the neck nodes. EGD showed the second complete remission and RUT was negative. She was diagnosed with stage IIA DLBLC and took three cycles of R-CHOP followed by radiotherapy of 3,600 cGr. In April 2007, DLBCL and gastric MALT lymphoma regressed completely. RUT was negative. In December 2008, DLBCL and gastric MALT lymphoma remained in complete remission state and RUT was negative. Until recently she has maintained complete remission state of DLBCL and gastric MALT lymphoma and negative RUT.
DISCUSSION
Three hypotheses have been suggested to explain high-grade lymphoma developing from the patients with low-grade MALT lymphoma. One is low-grade MALT lymphoma with large cell components.4 Usually low-grade MALT lymphoma can be regressed easily after H. pylori eradicaction and high-grade components may progress. In these cases, development of high-grade lymphoma may occur rapidly. Another is transformation of a low-grade lymphoma to a high-grade lymphoma.6,7 The other is that a completely new high-grade lymphoma can be developed from carcinogenic effect of the treatment for low-grade MALT lymphoma.8 Some chemotherapeutic agents or radiation have potent carcinogenic effect. It may need longer period than the other two situations. In our cases, the first case had taken 10 years to develop a high-grade lymphoma but there was no history of chemotherapy or radiotherapy. Therefore the second hypothesis seems the most suitable hypothesis. However, there is also the possibility that the gastric MALToma and the DLBLC in the cervical nodes were two unrelated phenomena. Iwano et al.4 reported three cases of rapidly developed high-grade lymphomas after H. pylori eradication for low-grade MALT lymphoma. They proposed that the first hypothesis is a possibility for their cases. Our second case can be explained by the same hypothesis.
Our study has a limitation of inconvenience by not obtaining molecular characteristics of our cases. Several studies have tried to elucidate the clonal progression of MALT lymphoma to DLBCL. However, the clonal relationship between MALT lymphoma cells and coexisting DLBCL cells is still controversial, and the exact mechanism of transition from MALT lymphoma to DLBCL remains unclear.7,9,10 A clonal linkage between small and large cell components in gastric DLBCL was shown by immunohistomchemistry genetic linkage of MALT lymphoma and de novo DLBCL.6,11 These findings suggest direct clonal progression from MALT lymphoma to DLBCL. However, clonal diversity has been reported, too.4,9 According to the hypothesis described above, we can guess that clonal diversity is a possibility by the first and the third hypotheses, while clonal homogeneity is also possible by the second hypothesis.
Starostik et al.12 proposed two pathways for the development and progression of gastric MALT lymphoma. T (11;18)(q21;q21)-positive lymphoma is characterized by a more advanced stage and unresponsiveness to H. pylori eradication and is unlikely to transform into a high-grade lymphoma.13,14 Whereas, t (11;18)(q21;q21)-negative lymphoma reveals more clonal aberrations and is prone to progress more frequently to DLBCL.15
The regression of gastric MALT lymphoma after the eradication of H. pylori was first reported by Wotherspoon et al.16 in 1933. Nowadays eradication therapy is thought to be the first-line treatment for patients with gastric MALT lymphoma. However, treatment strategy for patients without evidence of H. pylori infection has been controversial.1,17 More studies are required to clarify the effectiveness of antibiotic therapy for these subsets. In our first case, H. pylori on histology, H. pylori culture, RUT, UBT, and serology were all negative. Our patient without H. pylori infection took H. pylori eradication and then subtotal gastrectomy. In the past, surgical treatments were common for early stage gastric MALT lymphoma, resulting in about 90% of 5-year survival rates but a significant morbidity.18,19 Recent reports suggested that radiation therapy alone for localized H. pylori-negative or eradication resistant gastric MALT lymphoma is feasible and safe, and allows gastric preservation.2,20
Most localized gastric MALT lymphomas are cured well. But some of them suffer development to a high-grade DLBCL sooner or later. Furthermore, extra-abdominal development of DLBCLs can be possible without intra-abdominal relapse like our cases. Therefore, physicians should give attention to the development of DLBCLs in the intra-abdominal and extra-abdominal organs for a long period of time after achieving complete remission of low-grade MALT lymphomas.