INTRODUCTION
Superior mesenteric artery (SMA) syndrome, also known as Wilkie syndrome or cast syndrome, is an uncommon disease that results from SMA compression of the third portion of the duodenum.1 The diagnosis of SMA syndrome should be considered in a young underweight patient (i.e., low body mass index [BMI]) presenting with abdominal pain, nausea, anorexia, weight loss, and vomiting.2 Conventionally, the diagnosis is made using upper barium study or computed tomography (CT). To our knowledge, EUS has been used to diagnose SMA syndrome in a single case reported in the Korean literature,3 using a mini probe. No case studies of SMA syndrome have reported the development of this disorder in a patient with normal BMI. Therefore, we report a case of SMA syndrome diagnosed with the aid of linear endoscopic ultrasound (EUS) in a patient with normal BMI.
CASE REPORT
A 61-year-old female visited the emergency room complaining of sudden abdominal pain and nausea after overeating. She visited a local clinic where she underwent abdominal CT and was prescribed an analgesic, but the pain did not subside, so she visited our hospital. Her medical and social histories were noncontributory. At admission, her blood pressure was 120/80 mm Hg, pulse rate 68 per minute, respiratory rate 18 per minute, body temperature 36.9Ōäā, height 157 cm, weight 50 kg, and BMI 20.3. She denied a history of weight loss. The physical examination revealed an acutely ill-looking appearance with epigastric tenderness. The complete blood count showed leukocytes 10,600/┬ĄL, hemoglobin 13.3 g/dL, and platelet count 208,000/┬ĄL. The blood chemistry showed total bilirubin 0.5 mg/dL, aspartate aminotransferase 18 U/L, alanine aminotransferase 13 U/L, amylase 106 U/L, blood urea nitrogen 23 mg/dL, creatinine 0.67 mg/dL, and C-reactive protein 0.26 mg/dL.
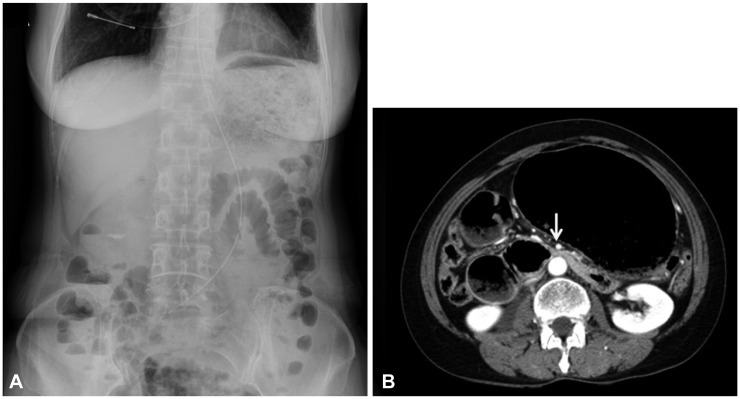
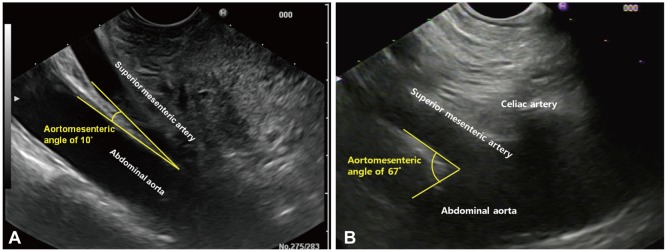
The initial abdominal X-ray revealed a large amount of food distending the stomach and mild gas accumulation in the small and large bowel (Fig. 1A). An abdominal CT taken at the local clinic showed a hugely distended stomach and duodenum (Fig. 1B). The aortomesenteric distance on CT was 7.4 mm which was decreased than normal mean aortomesenteric distance of 10 to 28 mm in healthy people.4 Conventional barium studies, which still play an important role for diagnosis of SMA syndrome, could not be performed because of severe gastroptosis and distended stomach. Then we performed esophagogastroduodenoscopy (EGD) and EUS to rule out intestinal intraluminal obstruction and gastric or duodenal ulcer disease that might be secondary to reflux or that might constitute a primary pathology mimicking SMA syndrome.5 EGD revealed a huge amount of food in the stomach (Fig. 2). Since that EUS could give us more information about surrounding organs which can directly compress the duodenum, we performed linear EUS (EG3870-UTK; Pentax, Tokyo, Japan). This did not show any abnormality of the gallbladder, common bile duct or pancreas. However, it showed the SMA running almost parallel to the abdominal aorta, with an aortomesenteric angle of 10┬░ (normal range, 38┬░ to 65┬░), confirming the diagnosis of SMA syndrome (Fig. 3, Supplementary Video 1 [available online at http://www.e-ce.org/]). The patient was managed conservatively with bowel rest and intravenous fluids. The abdominal pain and nausea were relieved and the patient started a soft diet on day 5 of admission and was discharged on day 7 of admission without any symptoms.
DISCUSSION
SMA syndrome is uncommon and characterized by postprandial epigastric pain, nausea, vomiting, anorexia, and weight loss.5 The symptoms of SMA syndrome are similar to those of small bowel obstruction, and include vomiting, nausea, early satiety, anorexia, and abdominal pain.6,7 The symptoms occur after or are aggravated by eating.4 SMA syndrome is favored by particular anatomic conditions, such as a short or hypertrophic ligament of Treitz, a low origin of the SMA, intestinal malrotation, and lumbar hyperlordosis.8 Undernutrition or rapid weight loss leads to reduced thickness of the adipose tissue in the aortomesenteric space.8
The following fluoroscopy-based radiological criteria are indicative of SMA syndrome: dilatation of the first and second parts of the duodenum with or without gastric dilatation, abrupt vertical or oblique compression of the third part of the duodenum, antiperistaltic waves of barium proximal to the obstruction, significant delay in gastroduodenal transit, and relief of the obstruction after postural changes.9 However, an upper barium study is of limited use for evaluation of SMA syndrome due to severe gastroptosis. The vascular abnormalities are well-delineated on fine-slice CT imaging with vascular reconstruction10 and measurement of the aorta-SMA angle.2 However, an upper barium study and CT involve considerable radiation exposure, especially in children or adolescents. The endoscopic approach minimizes the exposure to radiation, which might be of substantial benefit for young patients.4
The upper endoscopic findings of duodenal dilatation, stasis, and antiperistaltic waves suggest the presence of SMA syndrome. However, one must rule out other causes, especially duodenal obstruction. The diagnosis of SMA syndrome using upper endoscopy cannot always be made with certainty. According to the literature, endoscopy is of minor diagnostic value.11 However, upper gastrointestinal endoscopy should be performed to rule out intestinal intraluminal obstruction and gastric or duodenal ulcer disease that might be secondary to reflux or that might constitute a primary pathology, mimicking SMA syndrome.5 Although the endoscopic features of duodenal dilatation and stenosis are suggestive of SMA syndrome, other pathologies that present with features resembling those of SMA syndrome must be excluded.
The main anatomical feature of this syndrome is the narrowing of the angle between the SMA and the aorta, which is normally 38┬░ to 65┬░.2 In SMA syndrome, this angle is typically decreased to 6┬░ to 25┬░.12 The angle measured by linear EUS in our patient was 10┬░. The measurement of the aortomesenteric angle may be different according to the positional change. However, we believe that measurements have acceptable aortomesenteric angle differences among each position. The cause of this narrowing, other than congenital abnormality, is postulated to be related to the loss of intra-abdominal adipose tissue,2 which narrows the aorto-SMA angle, resulting in functional obstruction.2
Kim et al.3 reported the diagnosis of SMA syndrome using mini probe EUS. In the study, mini probe EUS was performed to evaluate the maximal aortomesenteric distance. In this study, upper gastrointestinal barium series to assess the diagnosis was limited by the presence of severe gastroptosis. Linear EUS provides a 120-degree sonographic image parallel to the axis of the echoendoscope. This linear orientation allows the measurement of the aortomesenteric angle. We measured the aortomesenteric angle in our study. The role of linear EUS in the diagnosis of SMA syndrome is unclear. Linear EUS can provide simultaneous endoscopic and ultrasonographic images of the stomach, duodenum, and surrounding structures. Recently, linear EUS was reported to be a useful diagnostic modality for evaluation of the aortomesenteric angle.13 In this context, linear EUS can confirm SMA syndrome without exposure to radiation or contrast medium. In addition, our patient showed X-ray findings of a ptotic, dilated stomach, which can prevent barium passage into the duodenum. In other words, an upper barium study might have been of limited use in our case.
Linear EUS imaging of the aortomesenteric angle can be obtained by scanning the abdominal aorta and SMA at the stomach position. After identifying the liver, clockwise rotation of the scope assists imaging of the abdominal aorta. The celiac artery and SMA are imaged when the scope is advanced caudally from this position along the abdominal aorta. However, the SMA is often difficult to image, particularly in SMA syndrome patients, due to artifacts related to retained food or air in the stomach. Because the scanning position of the mini probe is where the descending duodenal segment narrows,3,14 linear EUS from that position would assist imaging of the aortomesenteric angle. The assessment of aortomesenteric angle during deflation of the balloon appears to be reflective of accurate angle because the balloon inflation may cause narrowing of the angle by compression.
Individuals who are underweight or have lost weight are at increased risk of SMA syndrome.5 Interestingly, our patient had a normal BMI with no history of weight loss. The cause of her SMA syndrome is not known. There is a report of patients with a normal BMI and SMA syndrome, suggesting that low BMI is not necessary for its development.15 Weight loss does not appear to be responsible for all cases. For example, SMA syndrome is common following corrective spinal surgery for scoliosis.5 This procedure lengthens the spine cranially, displacing the SMA origin, decreasing the lateral mobility of the mesenteric artery, and reducing the acuteness of the aortomesenteric angle. The development of SMA syndrome may also be related to variation or distortion of the normal anatomy, such as short ligament of Treitz,5 low origin of the SMA,4 or esophagectomy.16 A case report of identical twins17 with SMA syndrome and another case in utero18 suggest a genetic predisposition in some patients. Therefore, anatomical variation or genetic susceptibility might have contributed to the SMA syndrome in our case. More case studies are needed to identify SMA syndrome with normal BMI.
SMA syndrome is a rare disease that presents with nonspecific symptoms that could delay diagnosis and treatment. Most patients present with upper gastrointestinal symptoms and are referred to gastroenterologists. This suggests that EGD with linear EUS could play an important role in the early diagnosis and treatment of SMA syndrome. Our case illustrates that SMA syndrome can occur in patients with normal BMI, although being underweight appears to predispose to its development. Although SMA syndrome is not typically within the scope of practice of endosonographers, it is useful to get familiar with the findings.