INTRODUCTION
Gut perforation during endoscopic retrograde cholangiopancreatography (ERCP) and stenting or due to migration of stents is a very rare complication, with an incidence of well below 2%. Moreover, direct duodenoscope-induced perforation of the lateral or medial duodenal wall is even less common, accounting for 0.1% of patients who undergo ERCP, but the perforation tends to be large and further away from the ampulla.1-6 Duodenal perforations from the superior duodenal angle to the descending duodenum may be complicated by leakage of digestive enzymes from the pancreas; this may severely damage other abdominal organs. Therefore, duodenal wall perforation is one of the most serious complications of ERCP.
Traditionally, the standard treatment for traumatic or iatrogenic duodenal perforation has been early surgical closure because of a relatively high mortality rate of 16% to 18%.1-4 However, although ERCP-related perforations of the lateral or medial duodenal wall usually require immediate surgery, some cases are inoperable because of the patient's advanced age and comorbidities that prohibit surgical treatment. For nonoperative management of a large perforation of the lateral or medial duodenal wall, early endoscopic closure of the perforation is essential.
TYPES OF IATROGENIC DUODENAL PERFORATIONS
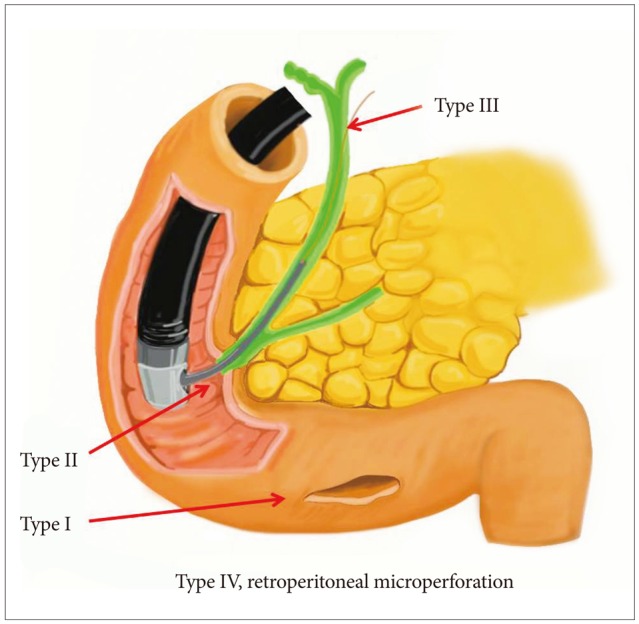
Several researchers have classified ERCP-related duodenal perforations according to the location or mechanism of injury and have recommended various treatments (Table 1, Fig. 1). Stapfer et al.1 classified perforations into four types, in decreasing order of severity, with implications for management. Type I perforations are perforations of the lateral or medial wall of the duodenum; these perforations involve large rents, are remote from the ampulla, and are caused by the endoscope itself or by the stent. These perforations cause considerable spillage, either retroperitoneally or intraperitoneally, necessitating aggressive surgical intervention. Type II perforations are perforations of the medial wall of the duodenum; these perforations are peri-Vaterian, are generally retroperitoneal, and occur during sphincterotomies. Type II perforations tend to be treated with conservative or minimally invasive therapies. Type III injuries are distal bile duct injuries caused by instrumentation and/or stenting in the proximity of an obstruction. These perforations are small and amenable to conservative management. Type IV perforations are tiny retroperitoneal perforations caused by the use of compressed air during endoscopy and can be managed conservatively. Howard et al.2 classified perforations into three types according to the mechanism of injury. Group I refers to guidewire-induced perforations; group II, periampullary perforations; and group III, duodenal perforations. Group III perforations require immediate surgery. Enns et al.3 suggested classification into three categories. Esophageal, gastric, and duodenal perforations require surgical management. Sphincterotomy-related perforation or guidewire-related perforation usually requires conservative management.
CLINICAL FEATURES
Known risk factors for ERCP-related duodenal perforation include old age, suspected dysfunction of the sphincter of Oddi, dilated bile duct, papillary stenosis, Billroth-II reconstruction, precut sphincterotomy, and long procedure duration.7-9 The classic presentation of duodenal perforation, with severe epigastric pain, vomiting, and epigastric tenderness progressing to generalized board-like rigidity, is seen only in a minority of cases. The symptoms and signs of ERCP-related perforations are often mild when this complication is recognized early.1,10-12 Therefore, the initial clinical presentation of patients with perforation is nonspecific. Duodenal perforation secondary to placement of a biliary endoprosthesis should be considered in all patients presenting with abdominal pain after such a placement. If a perforation is not recognized or suspected during ERCP, early diagnosis is difficult. Moreover, diagnosis is likely to be delayed if patients have concurrent elevated amylase level and the pain is attributed to post-ERCP pancreatitis. Clinical suspicion and diagnosis of a procedure-related perforation can be facilitated greatly by clinical findings and particularly by radiographic imaging with contrast studies, computed tomography (CT), and even magnetic resonance imaging. A multi-slice CT scan can provide an exact diagnosis of this complication.
ENDOSCOPIC MANAGEMENT
Duodenal wall perforation
Type I (perforation of lateral or medial duodenal wall; Stapfer et al.1) or group III (duodenal perforation remote from the papilla; Howard et al.2) injuries are usually large and traditionally require immediate surgery for repair after prompt diagnosis. In a study by Stapfer et al.,1 surgery was recommended for patients with the following criteria: large contrast extravasation on ERCP; contrast-enhanced CT scans showing intraperitoneal or retroperitoneal fluid collection; massive subcutaneous emphysema or suspected perforation in association with retained material (i.e., stones or ERCP wire/basket). In cases of peri-Vaterian injuries, they suggested conservative management with serial radiographic examination. Howard et al.2 also suggested the use of endoscopic drainage to divert the bile, pancreatic, and/or duodenal fluids away from the perforation, and showed that the endoscopic approach reduced the rates of surgery and mortality, and the length of hospital stay. Unlike more common spontaneous perforations resulting from peptic ulcer disease, endoscopic therapy-related iatrogenic perforations have a relatively lower chance of bacterial contamination in a fasting state; therefore, these patients can occasionally be managed using nonsurgical means such as endoscopic therapies. A small amount of bacterial contamination may be controlled by conservative managements, including nil by mouth, nasogastric or nasoduodenal drainage, intensive fluid therapy, and antibiotics.13
Trials of endoscopic management have recently been performed and endorsed. There have been sporadic reports on the use of an endoscopic clipping device for the closure of iatrogenic perforation during endoscopic mucosal resection (EMR), sphincterotomy, or stent migration in the esophagus, stomach, and duodenum.14-19 Although surgery remains the standard treatment for duodenal perforations caused by the endoscope itself, the outcomes from case reports support the beneficial role of endoclips in the closure of these defects.5,20-22 In particular, some reports have indicated that nonsurgical treatment is possible for perforation of the upper gastrointestinal tract when peritonitis remains localized. The clinician's familiarity with endoclips and the immediate availability and proper use of endoclips may help to avoid surgery for a selected group of patients with a high surgical risk.5
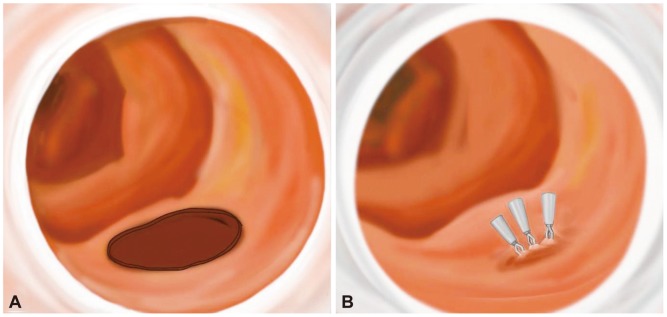
Typical primary endoscopic closure by endoclips may be difficult because of the position of the lateral wall and the complexity of aligning the perforation with the endoscope. To approximate the perforated hole and adherent hemoclips, glue injection and spraying may be successfully performed under cap-fitted endoscopy (Figs. 2, 3).18,20 Glue injection may cause multiple clips to adhere to each other and provide a cushion effect below the base of the perforation. In addition, since clipping with a duodenoscopy can be technically difficult, a cap-assisted, forward-view endoscopy can be used effectively. The cap-assisted endoscopy method under direct visualization through a transparent hood may be helpful in reducing the manipulation time of the procedure by allowing a good visual field and ensuring the safety margin during clipping.5
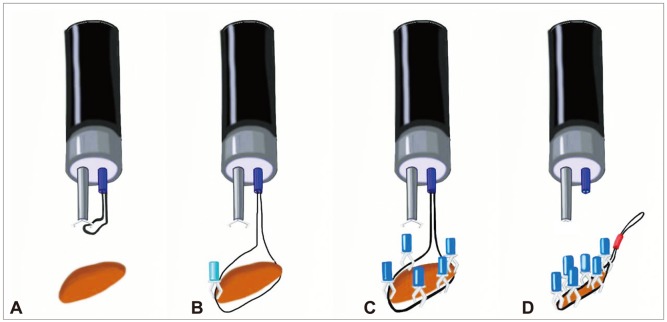
However, endoscopic repair using endoclips can be limited in large perforations or in those with tangential angles. A wide perforation is difficult to close because of slippage of the perforation edge from the clip while the clip is maneuvered across the defect to grasp the opposite edge of the perforation. Everted perforation edges also make it impossible to grasp the tissue with endoclips. As a modified method without additional glue injection, endoscopic closure using endoloops with endoclips has provided good clinical results, even though it has been described only in case reports.23-25 Clinical trials have been performed using an endoloop and multiple hemoclips to cover large mucosal defects after endoscopic submucosal dissection or ERCP-induced perforation.23-28 After placement of the endoloop around the perforated area through 1- or 2-channel endoscopy, multiple clips are attached with the endoloop to the perforated area; finally, the endoloop is tightened, and this closes the perforated area (Figs. 4, 5).29,30
Technically, endoscopic band ligation (EBL) is a simple procedure to treat bleeding from esophageal varices. The simplicity of the technique and low complication rates compared with those of sclerotherapy have contributed to its growing popularity.5 EBL has also been widely used in the management of non-variceal hemorrhage from Dieulafoy's ulcer, gastric angiodysplasia, and polypectomy-induced bleeding. In addition, several reports have described the use of EBL in rectal and duodenal perforations during EMR. Theoretically, EBL can readily approximate both edges of the perforation. Therefore, complete sutures to the remaining wall by additional bands or endoclips may be simple to achieve, even for a large perforation (Fig. 6). EBL can also reduce the procedure time in comparison with that required for clipping. Immediate closure can prevent the need for surgery or the development of serious peritonitis caused by leakage of gastric contents. Finally, the use of additional clips to suture the perforation after EBL might not be necessary.17 Thus, EBL may be also adopted for duodenoscope-induced duodenal wall perforation. However, more experimental studies are needed to confirm its suitability.
Finally, recent studies have reported high technical success rates for primary closure of an acute iatrogenic perforation with newly developed clips or endoscopic suturing devices developed for NOTES.5,13-16 These devices include through the scope (TTS) clips, such as the QuickClip 2 (Olympus Inc., Center Valley, PA, USA), the Resolution clip (Boston Scientific Inc., Natick, MA, USA), and the Tri-Clip and Instinct clip (Cook Medical, Winston-Salem, NC, USA); the over the scope clip (OTSC) system (Ovesco Endoscopy AG, Tubingen, Germany); and endoscopic suturing devices such as T tags (Ethicon Endo-Surgery, Cincinnati, OH, USA) and the flexible Endo Stitch (Covidien, Mansfield, MA, USA). Closure of luminal perforations >20 mm in size may be difficult. For larger gastric defects, TTS clips can be placed around the circumference of the perforation and lassoed together with a detachable plastic snare (Endo-loop; Olympus Inc.).17 Among the newly developed devices, OTSC has been used for the closure of perforation <20 mm in size, and ex vivo studies have shown that colon defects measuring 10 to 30 mm can be closed with a single OTSC.16 However, although some techniques have been developed to correct deficits in clip placement, they are not commonly practiced. Some of these devices may prove suitable for the closure of defects throughout the intestinal tract, but their use is limited by the endoscopist's experience, device availability, and cost. Currently, no one particular technique has demonstrated proven efficacy or greater reliability over other closure modalities.
Stent-induced perforation
Gut perforation has been reported following migration of all types of stents, irrespective of material, make, mode of placement, caliber, or size. The flexibility of the stent might play a role in influencing stent migration.10-12,31-36 None of the stents in current use have been implicated in particular. However, a straight-type stent with low flexibility may increase migration, especially in sphincterotomy state. For duodenal perforation secondary to placement of a biliary endoprosthesis, the treatment of choice is endoscopic removal of the endoprosthesis in the case of a localized process, and then the perforation can be su tured by a simple closure with hemoclips or hemoclips with glue injection.18 However, surgery should be reserved for patie nts with peritonitis or extensive retroperitoneal phlegmon.35,36
Sphincterotomy-induced perforation
The management of perforations after therapeutic periampullary endoscopic procedures has remained controversial. If perforation is suspected, prompt diagnosis and initiation of systemic support with broad-spectrum antibiotics and intravenous resuscitation is mandatory. Asymptomatic patients with evidence of retroperitoneal free air alone can normally be treated with conservative management. When perforation is confirmed, external/internal biliary, duodenal, and/or pancreatic drainage may be indicated.26 Some patients can be managed by diversion or decompression of contents from the duodenum by placing a nasoduodenal tube alone or with internal pancreaticobiliary drainage. Another option is diversion: a fully covered self-expandable metal stent can be used for the treatment of biliary sphincterotomy-induced perforation.27,28 However, inappropriate biliary drainage may cause bile infiltration or fluid leakage into the perforated site, thus increasing morbidity.1 If patients develop abdominal pain or fever or show clinical signs of toxicity, surgical consultation should be obtained, and operative exploration for effective repair or drainage should be considered, especially in elderly people or otherwise chronically ill patients less able to withstand physiologic stress.
Guidewire-induced perforation
Guidewire-induced perforation is defined as the entrance of the guidewire into the retroperitoneal space or through the bile duct during cannulation attempts or attempts to pass a stricture. These perforations may be also associated with injection of contrast into the retroperitoneal space.37 They are often very small in size and usually have no clinical consequences. Therefore, most cases of guidewire-induced perforation can be treated with conservative management, as mentioned above for sphincterotomy-induced perforations, or temporary diversion of the bile duct by a plastic or fully covered metallic stent.
CONCLUSIONS
In summary, for the successful management of ERCP-related duodenal perforations, prompt recognition and rapid management, including endoscopic closure or surgery, are the key determinants for lessening the significant morbidity and mortality. Delayed diagnosis and surgery are associated with a high mortality rate. Until recently, immediate surgical closure has been the standard treatment for traumatic or iatrogenic duodenal wall perforation, with the exception of guidewire-induced perforations or peri-Vaterian injuries. Recent endoscopic trials using clips, endoloops, glue injection, and newly developed devices such as OTSCs for the management of duodenal perforation have reported successful outcomes, although there is no clear consensus for primary repair because of the limited number of cases. Although nonsurgical suturing therapies are not widely accepted yet as the primary treatment for ERCP-related duodenal wall perforation, endos copic indications may be broadened in selected cases with the de velopment of endoscopic skills and devices. In addition, with im provement in endoscopic devices, endoscopic treatment may replace surgical management in the near future.