INTRODUCTION
Although primary gastric lymphoma accounts for only 1% to 7% of all gastric malignant tumors, it constitutes approximately 60% of lymphomas of the gastrointestinal tract.1,2 Gastric lymphoma occurs primarily in middle-aged patients (age, 40 to 60 years), with no sex predilection.1-3 Since Isaacson and Wright4 first established the concept of lymphoma originating from mucosa-associated lymphoid tissue (MALT) in 1983, the term MALT lymphoma has been widely accepted. Recently, MALT lymphoma was reclassified as extranodal marginal zone B-cell lymphoma of MALT type by the World Health Organization.5 Numerous studies investigating the treatment of gastric MALT lymphoma have been published to date. Currently, eradication of Helicobacter pylori is recommended as the initial treatment of choice, with a cure rate ranging from 60% to 80%.3,6 Patients who are unresponsive to H. pylori eradication are indicated for second-line therapy. Treatment options for refractory patients include surgery, radiation therapy, and chemotherapy, with radiation and chemotherapy being the preferred treatments.7-10 However, there is no consensus on the second-line therapy. Here, we report the case of a female patient with gastric MALT lymphoma unresponsive to H. pylori eradication, which was successfully treated by gastric polypectomy.
CASE REPORT
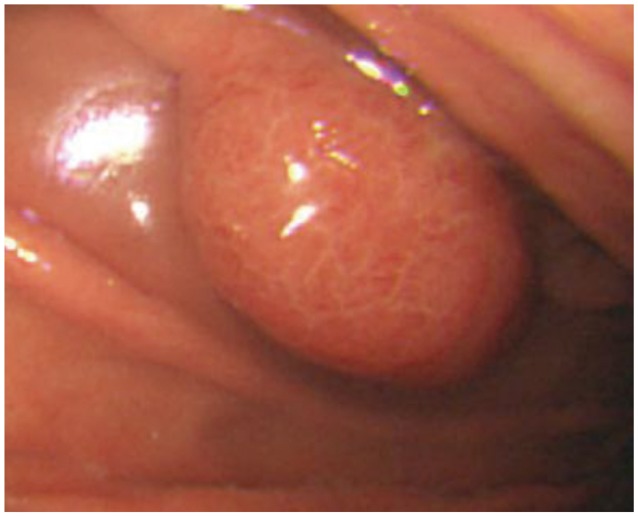
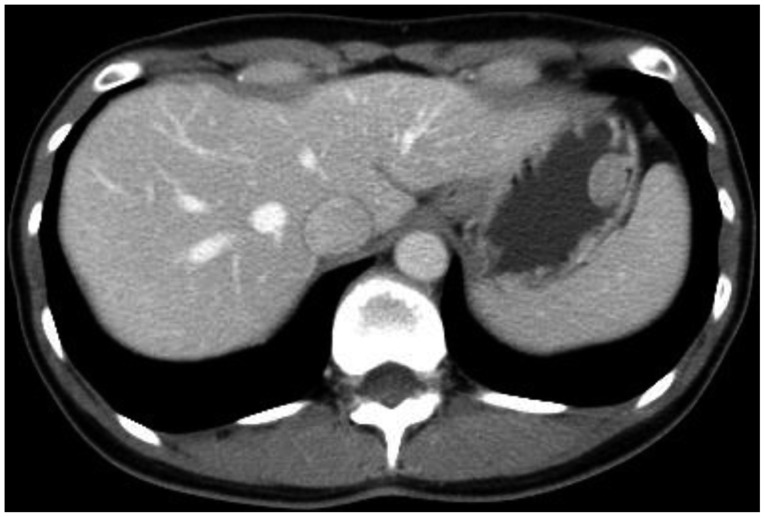
A 34-year-old woman visited our hospital for the management of a gastric polyp detected by an upper gastrointestinal endoscopy recently performed at a private clinic during a routine medical examination. She had no record of medical illness other than a history of uterine myomectomy at our hospital. She was asymptomatic, and her physical examination did not reveal any abnormality. She underwent upper gastrointestinal endoscopy, and a 2.5-cm polypoid lesion was found on the great curvature side of the upper body of the stomach (Fig. 1). Biopsy was performed. The pathologic review was suggestive of early-stage marginal zone B-cell lymphoma of MALT type. The tissue was also positive for H. pylori on Giemsa staining. Her laboratory data were as follows: white blood cells, 7,710/mm3 (neutrophil 58.7%, lymphocyte 32.6%, monocyte 5.5%); hemoglobin, 14.1 g/dL; platelets, 176,000/mm3; total protein, 6.1 g/dL; albumin, 3.7 g/dL; total bilirubin, 0.7 mg/dL; aspartate aminotransferase, 20 IU/L; alanine aminotransferase, 21 IU/L; alkaline phosphatase, 41 IU/L; blood urea nitrogen, 11.4 mg/dL; creatinine, 0.83 mg/dL; sodium, 143 mEq/L; potassium, 4.0 mEq/L; chloride, 107 mEq/L; and lactate dehydrogenase, 304 IU/L. An abdominal computed tomography (CT) showed a 1.8-cm polypoid lesion with minimal stalk in the high body of the stomach. No other abnormal findings, including lymph node enlargement, were noted (Fig. 2). Bone marrow examination revealed no abnormal finding.
As the initial treatment, H. pylori eradication was performed with a 2-week course of amoxicillin (2├Ś1,000 mg daily), clarithromycin (2├Ś500 mg daily), and rabeprazole (10 mg daily). The eradication was successful, as confirmed by Giemsa staining in the follow-up endoscopy. However, on endoscopy 6 months later, there was no change in the size and morphology of the polypoid MALT lymphoma lesion. Multiple specimens were obtained, and the pathologic evaluation showed the same result of marginal zone B-cell lymphoma of MALT type. We also performed abdominal CT and positron emission tomography but found no systemic lymphoma involvement.
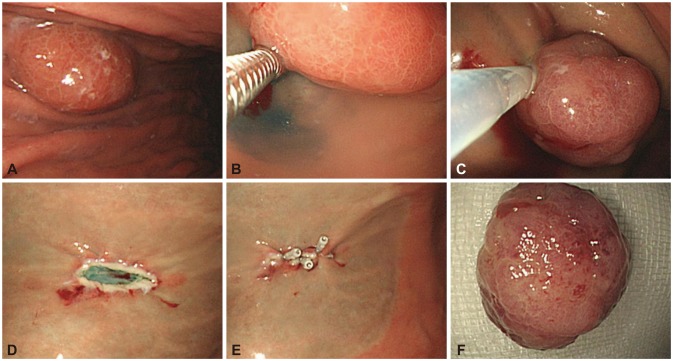
To confirm the diagnosis of low-grade lymphoma and exclude the possibility of high-grade lymphoma, we decided to perform polypectomy, which allows collection of enough tissue specimens including the submucosal layer. The lymphoma was removed en bloc, endoscopically with a snare (Fig. 3), after submucosal injection of hypertonic saline. No complication occurred. The final pathologic diagnosis was marginal zone B-cell lymphoma of MALT type, polypoid (Fig. 4). The lymphoma was 1.8├Ś1.3 cm in size and confined to the submucosal layer. The lateral and basal resection margin was clear. At follow-up endoscopy 6 months after polypectomy, a linear whitish scar was observed without evidence of recurrence (Fig. 5). She has been followed up for 3 years without recurrence.
DISCUSSION
The most common endoscopic feature of gastric MALT lymphoma is ulcer or erosion, which accounts for >40% of gastric MALT lymphoma cases. Mass or polyp is observed in 7% to 25% of the patients.3,11,12 Gastric MALT lymphoma may develop at any site of the stomach.3,11,12 The diagnosis of lymphoma is confirmed by pathologic evaluation of tissue. However, detection of a lymphoma confined to the submucosal layer by conventional endoscopic biopsy is difficult.13 An accurate diagnosis of low-grade and high-grade lymphoma was obtained on initial endoscopic biopsy in 75% and 79% cases, respectively.14 Endoscopic mucosal resection (EMR) can be used for accurate diagnosis. Suekane et al.15 recommend performing EMR in patients with suspected early-stage lymphoma who were not diagnosed by conventional endoscopic biopsy.
H. pylori eradication is the recommended treatment of choice for gastric MALT lymphoma. However, failure of H. pylori eradication was noted in patients with submucosal invasion, high-grade lymphoma, or involvement of perigastric lymph nodes.16-18 These patients are subject to secondary treatment. However, at present, there is no consensus on the standard approach for treating patients refractory to H. pylori eradication. In one multicenter study, the 5-year survival rate of surgically and nonsurgically treated patients with stage I or II gastric MALT lymphoma was 84.4% and 82%, respectively, with no statistical difference between the groups.10 Suekane et al.15 evaluated the role of radiation therapy and suggested that lowdose radiation therapy within 4 weeks was effective for gastric MALT lymphoma. Yoon and Hochberg9 reported that among patients with stage I or II gastric MALT lymphoma treated with surgery, radiation therapy, and chemotherapy, those treated with chemotherapy achieved the lowest recurrence rate and the highest 10-year survival rate.
EMR is a useful technique in the tissue diagnosis of gastric MALT lymphoma. However, evidence on the efficacy of gastric polypectomy for the treatment of refractory MALT lymphoma is not well established. A few cases of EMR used as a curative method in patients who were unable or unwilling to undergo a radical gastrectomy can be found in the literature. Seifert et al.13 reported the treatment outcome of 420 patients with gastric MALT lymphoma, and only one patient underwent a polypectomy as a second-line treatment after failing to achieve complete remission by H. pylori eradication. Youn et al.19 reported one patient with nodular gastric MALT lymphoma treated with polypectomy as a primary therapy who survived for 20 months after treatment. However, randomized trials with long-term follow-up periods seem to be required to evaluate the efficacy of mucosal resection as a curative modality, although such studies would be difficult because of the small number of cases.
Here, we report the case of a patient with gastric MALT lymphoma who was successfully treated with gastric polypectomy without recurrence in the 3-year follow-up period.