AbstractBackground/AimsTo evaluate the yields and utility of 19-gauge (G) Trucut biopsy (TCB) versus 22 G fine needle aspiration (FNA) for diagnosing gastric subepithelial tumors (SETs).
MethodsWe retrieved data for 152 patients with a gastric SET larger than 2 cm who had undergone endoscopic ultrasonography (EUS)-guided 19 G TCB (n=90) or 22 G FNA (n=62). Relevant clinical, tumor-specific, and EUS procedural information was reviewed retrospectively.
ResultsA specific diagnosis was made for 76 gastrointestinal stromal tumors (GISTs) and 51 non-GIST SETs. The diagnostic yield of TCB was greater than that of FNA (77.8% vs. 38.7%, p<0.001). The percentage of non-diagnostic specimens (suspicious and insufficient) was significantly lower in the TCB group (6.7% and 15.5%, respectively) than in the FNA group (22.6% and 38.7%, respectively; both p<0.001). TCB accurately diagnosed 90.9% of GISTs and 81.1% of non-GIST SETs, whereas FNA accurately diagnosed 68.8% of GISTs and 14.3% of non-GIST SETs. There were nine technical failures with TCB, and the rate of adverse events did not differ between the groups (TCB vs. FNA, 3.3% vs. 8.1%; p=0.27).
INTRODUCTIONEndoscopic ultrasonography (EUS) is useful for evaluating gastric subepithelial tumors (SETs). EUS allows the differentiation of intramural lesions from extrinsic compression and characterization of the tumors by providing information about the layer of origin, echo pattern, and margin. However, because it is impossible to differentiate definitely between benign and malignant SETs by EUS alone, histologic or cytologic confirmation is necessary.1,2,3
EUS-guided sampling via fine-needle aspiration (FNA) is commonly used to obtain specimens from lesions within the gastrointestinal (GI) tract and adjacent organs.4,5 Sufficient diagnostic yields ranging from 60% to 90% have been reported using 22-gauge (G) needle FNA with an excellent safety profile.1,6,7,8 However, FNA is limited in that the aspirates consist mostly of cellular materials, and thus, specimens are not always available to assess architectural details or perform immunohistochemical analysis.9 In addition, its utility is influenced by several factors such as the presence of onsite cytopathologists, the number of passes, the use of a stylet or suction during FNA, and special techniques to procure better quality specimens.10,11
Cutting biopsy with a 19 G Trucut biopsy (TCB) was introduced to overcome these limitations of FNA in 2002.12 EUS-TCB provides larger core tissue specimens that preserve the tissue architecture and allow histological examination with immunohistochemistry, which is especially useful in certain tumors such as lymphomas and gastrointestinal stromal tumors (GISTs). Nevertheless, the range of diagnostic yields for TCB has been reported as 47% to 78% in various organs.13,14,15 These less than expected results of TCB are related to technical difficulties associated with the use of a large, stiff 19 G needle. In particular, TCB displayed extremely high failure rates when the transduodenal approach was employed.16 Complications are more frequently encountered with TCB in pancreatic diseases.11,17 However, the details of TCB-related technical failure and complications in gastric SETs are lacking.
Hence, the aim of this study was to compare the yields and utility of 19 G TCB versus 22 G FNA with a large volume of serial cases of gastric SETs larger than 2 cm in diameter.
MATERIALS AND METHODSStudy populationThe records of 184 patients with a gastric SET larger than 2 cm in diameter who underwent EUS-guided needle sampling between November 2005 and May 2012 at Asan Medical Center in Korea were retrieved from a consecutively collected database. A gastric SET larger than 2 cm without benign EUS features, such as a typical lipoma, cystic tumor, or ectopic pancreas, was subject to EUS-guided needle sampling for pathologic diagnosis.18,19,20 Thirty-two patients who underwent EUS-guided sampling with other needle types were excluded from the analysis, including five, three, and 24 patients who underwent 25 G FNA, 22 G TCB, and 19 G FNA, respectively. Thus, a total of 152 patients were included in this study. Tumor size, gastric location, EUS echo features, number of needle passes, and procedure-related adverse events were reviewed. The diagnostic yields of FNA and TCB were assessed by definitive cytopathology obtained from aspiration smears and/or cell blocks, core biopsies, and endoscopic or surgical resection.
All patients provided written informed consent before their procedure. The study protocol was approved by the Institutional Review Board of the Asan Medical Center.
EUS proceduresEUS-guided FNA or TCB was performed using a linear-array echoendoscope (GF-UCT2000-OL5; Olympus, Tokyo, Japan) with patients under conscious sedation. The procedures were performed by seven endoscopists who were specialists in diagnostic EUS of the upper GI tract. TCB was introduced as an alternative sampling method in January 2007 in our endoscopic unit. The needle type was decided by an operating endosonographer. The yearly usages of 22 G FNA and19 G TCB during the study period were as follows: 2 vs. 0 in 2005 (from November), 5 vs. 0 in 2006, 2 vs. 2 in 2007, 2 vs. 9 in 2008, 8 vs. 15 in 2009, 16 vs. 24 in 2010, 17 vs. 26 in 2011, and 10 vs. 14 in 2012 (until May). For the aspiration procedure with a 22 G FNA needle (Echotip; Wilson-Cook, Winston-Salem, NC, USA), negative pressure was applied using a 10-mL syringe if the preceding attempt did not yield aspirate.21 Aspirate was placed onto glass slides and assessed macroscopically by endosonographers. Particles of aspirate were collected into a formalin solution for cell block preparation (hematoxylin and eosin stain) and subsequent immunohistochemical staining. Some aspirated samples were smeared onto glass slides and fixed immediately in 95% absolute alcohol for cytologic examination (Papanicolaou staining). For the TCB technique, a 19 G needle (Quick-Core; Wilson-Cook) was inserted into a lesion and withdrawn after the spring-loaded outer needle was fired.20 Core biopsy specimens were retrieved from the 20-mm tissue tray and fixed in formalin. Additional cytologic examination was not performed for the specimens acquired by TCB. An on-site cytopathologist was not available during the procedure.
Diagnostic definitionsPathologic results were categorized as diagnostic or nondiagnostic. A diagnostic result was defined when sufficient samples were obtained to allow for the architectural details and/or a specific diagnosis to be established by immunohistochemistry including stains for c-kit (CD117), CD34, smooth muscle actin, and S100. A nondiagnostic result was defined as follows: (1) suspicious, a needle sample from which cellular materials was obtained, but the quantity was not sufficient for architectural details or immunohistochemistry; or (2) insufficient, a scant sample from which a diagnosis could not be made. A diagnosis of GIST was made when microscopy revealed spindle or epithelioid cells that were positive for c-kit with or without positive CD34 immunohistochemistry.22 Leiomyoma was typified by smooth muscle or spindle cells that were positive for actin and negative for c-kit. A tumor of neural origin (schwannoma) was defined by the presence of spindle cells that were positive for S100.
Statistical analysisDescriptive statistics were used to characterize the study subjects. Continuous variables were expressed as means┬▒SD and compared using Student t-test. The chi-square test was used to compare categorical variables. All statistical calculations were 2-sided, and a p-value less than 0.05 indicated statistical significance. Statistical analyses were performed using SPSS version 20 (IBM Co., Armonk, NY, USA).
RESULTSOf the 152 study patients, 62 (40.8%) underwent 22 G FNA, and 90 patients (59.2%) underwent 19 G TCB (Fig. 1). EUS-guided sampling was diagnostic for 94 lesions (61.8%) and nondiagnostic for 58 lesions (38.2%). A pathologic diagnosis was made additionally by surgical resection in 83 patients (54.6%) and by endoscopic resection in seven patients (4.6%). The final pathology could not be determined in 25 patients as follows: medical follow-up with a suspicion of benign lesion on EUS and/or sampling specimens in 18 patients, severe comorbidity for operation in two patients, and refusal to undergo resection in five patients. Of the 127 identified gastric SETs, 76 lesions (59.8%) were found to be GISTs. The final pathology of gastric SETs that were sampled by 22 G FNA and 19 G TCB is summarized in Table 1 (p=0.008).
The clinical and EUS features of patients who underwent EUS-guided sampling because of SET are shown in Table 2. The mean age of patients was 55.5 years, and 50% were male. The mean tumor size was 40.5 mm, and 26 tumors (17.1%) were larger than 5 cm in diameter. There was no difference in diameter between the 22 G FNA and 19 G TCB groups (37.4┬▒23.6 and 42.6┬▒32.9 mm, respectively; p=0.23). For lower third lesions, FNA and TCB were applied in 16 and seven patients, respectively (25.8% vs. 7.8%, p=0.003). On EUS, internal echo-textures of heterogeneity, cystic foci, and hyperechoic spots were similar between the groups. Irregularity of the outer border was found more frequently in tumors sampled using 19 G TCB. A median of three needle passes was required for each target lesion (p=0.33). Technical failure of TCB was reported in nine patients, who were converted to FNA. The locations of these lesions consisted of the cardia (one case), greater curvature side of the fundus (two cases), lesser curvature side of the body (four cases), and greater curvature side of the antrum (two cases). Procedure-related adverse events were reported in eight patients (5.3%; FNA vs. TCB, 8.1% vs. 3.3%; p=0.27). All adverse events improved during the 24-hour postprocedure hospital stay. One case of TCB-related abdominal pain resolved spontaneously, and two cases of immediate bleeding after TCB puncture were managed with endoscopic hemostasis.
The overall diagnostic yield of EUS-guided needle sampling was 61.8% (Fig. 2). The diagnostic yield of 19 G TCB was significantly higher than that of 22 G FNA (77.8% vs. 38.7%, p<0.001). The percentages of nondiagnostic specimens (suspicious and insufficient) were significantly lower for TCB (6.7% and 15.5%, respectively) than for FNA (22.6% and 38.7%, respectively). Of 76 GISTs, diagnostic specimens were obtained for 90.9% of tumors using TCB and 68.8% of tumors using FNA (p=0.02) (Fig. 3). Of 51 non-GIST SETs, the diagnostic yields were 81.1% using TCB and 14.3% using FNA (p<0.001).
DISCUSSIONThis study demonstrated that the yield of 19 G TCB was significantly higher than that of 22 G FNA for diagnosing gastric SETs larger than 2 cm. The complication rate of TCB was similar to that of FNA; however, technical failure occurred in the use of TCB.
Because cutting biopsy provides more reliable specimens for architectural detail and immunohistochemistry, TCB has been recommended for the histologic diagnosis of SET. However, the diagnostic yields of TCB were reported to range 47% to 78%, and inadequate samples were obtained in some proportion of cases.18 One of the reasons for these variously estimated yields may be explained by procedural difficulties of TCB using stiff, large-bore 19 G needles, especially in angular positions. In a study with SETs greater than 2 cm in the stomach, the diagnostic yield of TCB was 63%, and tumor tissue was insufficient in 30.6% of punctured cases.15 They reported that a tumor location on the lesser curvature of the stomach was the only independent predictor of obtaining diagnostic material, and failure of needle puncture occurred in 6.1% of tumors in an unfavorable location. By contrast, TCB allowed core specimens to be obtained more frequently for histologic assessment than FNA (95.3% vs. 27.8%) and displayed greater diagnostic accuracy (88.3% vs. 77.8%) in lesions without angulation.23 Previously, we also reported that 13.8% of gastric SETs were not readily accessible using 19 G TCB.20 In the present study, 19 G TCB provided diagnostic specimens for 77.8% of gastric SETs, and the rates of non-diagnostic suspicious or insufficient samples obtained by TCB were significantly lower than those for FNA.
The overall diagnostic yield of 22 G FNA in our study was lower than that reported.18 This is probably explained by the absence of an on-site cytopathologist, leading to a suboptimal evaluation of FNA aspirates.10 In a study assessing the quality of EUS-FNA performed by 92 endosonographers, they explained the lower sensitivity of FNA by the unavailability of rapid on-site cytopathological evaluation.23 In addition, on-site cytopathological evaluation increased the adequacy rates of FNA specimens by 10% to 29%.6,24,25
Between 10% and 20% of gastric SETs are malignant or potentially malignant, and GISTs are the most predominate tumors.26 Studies of EUS-guided needle sampling have been focused on the differentiation of GISTs from other benign tumors. In our study, 19 G TCB displayed significantly higher diagnostic yields for both GISTs (90.9%) and non-GIST SETs (81.1%). The positive yield rate with 22 G FNA for gastric GISTs was higher (68.8%) than that for all gastric SETs (38.7%) and non-GIST SETs (14.3%). We speculate that the cytologic diagnosis of GIST based on c-kit-positive immunostaining might be responsible for the higher yield of FNA. A malignant or potentially malignant GIST has higher cellularity and mitotic features, and thus, substantial amounts of cells might be aspirated into the FNA needle. Likewise, the diagnostic yield of 22 G FNA for GISTs has been demonstrated to range from 74% to 100%.1,7,13 The diagnostic yields were estimated to be as high as 91% for malignant GISTs, and the accuracy was 94.2% for identifying potentially malignant SETs.1,27
A few studies reported TCB-related adverse event rates of 2% to 4%.15,28 These events included fever, localized abdominal pain, puncture site infection, and bleeding.8,28,29 We found a TCB-related adverse event rate of 3.3%, which was comparable to that of 22 G FNA, and all adverse events were manageable.
Technical failure of TCB occurred in nine of 99 attempts (9.1%), and these patients were converted to FNA. Technical failure of TCB is caused by the needle, which is stiff and poorly maneuverable, and therefore, it fails to approach the lesion in some locations.11 Recently, to overcome this limitation, a new generation of core biopsy devices of various sizes (ProCore; Cook Medical) were introduced.30 Several studies evaluated the performance of ProCore at various organs (pancreatic masses, lymph nodes, and other indications).31,32,33 However, they reported inconclusive data, and well-designed randomized trials comparing the different gauge ProCore and standard FNA needles for gastric SETs are required.31
This study is subject to the limitations inherent in retrospective designs with observation data and represents the experience of a single tertiary referral center, and our findings might not be generalizable. Mostly, there should be a special concern of selection bias for the diagnostic yields of EUS-guided sampling. As needle types were selected by operating endosonographers, the yearly usages of needles (FNA vs. TCB) were different. In total, 77% of EUS-FNA procedures were performed before 2010, and 55% of EUS-TCB procedures were performed before 2010, which means that EUS-TCB was performed after endosonographers obtained sufficient experience in EUS-guided tissue acquisition. Presumptive EUS diagnosis and the needle selection by individual endosonographers may affect the diagnostic yields. There might be a low probability of diagnostic discordance because not all EUS-guided diagnoses could be reviewed with reference to surgical pathology. In addition, it is difficult to determine whether the difference in the diagnostic yields of the two sampling methods arose from the techniques (biopsy or aspiration) or the size of the needle (19 G vs. 22 G). However, we believe that this lack of clarity merits a future comprehensive study to improve the diagnostic yields of EUS-guided needle sampling for gastric SETs.
In conclusion, 19 G TCB is safe and highly valuable for diagnosing gastric SETs larger than 2 cm if technical failure can be avoided.
References1. Ando N, Goto H, Niwa Y, et al. The diagnosis of GI stromal tumors with EUS-guided fine needle aspiration with immunohistochemical analysis. Gastrointest Endosc 2002;55:37ŌĆō43. 11756912.
2. Wiech T, Walch A, Werner M. Histopathological classification of nonneoplastic and neoplastic gastrointestinal submucosal lesions. Endoscopy 2005;37:630ŌĆō634. 16010607.
3. Papanikolaou IS, Triantafyllou K, Kourikou A, R├Čsch T. Endoscopic ultrasonography for gastric submucosal lesions. World J Gastrointest Endosc 2011;3:86ŌĆō94. 21772939.
5. Mortensen MB, Pless T, Durup J, Ainsworth AP, Plagborg GJ, Hovendal C. Clinical impact of endoscopic ultrasound-guided fine needle aspiration biopsy in patients with upper gastrointestinal tract malignancies. A prospective study. Endoscopy 2001;33:478ŌĆō483. 11437039.
6. Chang KJ, Katz KD, Durbin TE, et al. Endoscopic ultrasound-guided fine-needle aspiration. Gastrointest Endosc 1994;40:694ŌĆō699. 7859967.
7. Sepe PS, Moparty B, Pitman MB, Saltzman JR, Brugge WR. EUS-guided FNA for the diagnosis of GI stromal cell tumors: sensitivity and cytologic yield. Gastrointest Endosc 2009;70:254ŌĆō261. 19482280.
8. Varadarajulu S, Fraig M, Schmulewitz N, et al. Comparison of EUS-guided 19-gauge Trucut needle biopsy with EUS-guided fine-needle aspiration. Endoscopy 2004;36:397ŌĆō401. 15100946.
9. Rader AE, Avery A, Wait CL, McGreevey LS, Faigel D, Heinrich MC. Fine-needle aspiration biopsy diagnosis of gastrointestinal stromal tumors using morphology, immunocytochemistry, and mutational analysis of c-kit. Cancer 2001;93:269ŌĆō275. 11507701.
10. Ramesh J, Varadarajulu S. How can we get the best results with endoscopic ultrasound-guided fine needle aspiration? Clin Endosc 2012;45:132ŌĆō137. 22866253.
11. Karadsheh Z, Al-Haddad M. Endoscopic ultrasound guided fine needle tissue acquisition: where we stand in 2013? World J Gastroenterol 2014;20:2176ŌĆō2185. 24605016.
12. Wiersema MJ, Levy MJ, Harewood GC, Vazquez-Sequeiros E, Jondal ML, Wiersema LM. Initial experience with EUS-guided trucut needle biopsies of perigastric organs. Gastrointest Endosc 2002;56:275ŌĆō278. 12145612.
13. Fern├Īndez-Esparrach G, Sendino O, Sol├® M, et al. Endoscopic ultrasound-guided fine-needle aspiration and trucut biopsy in the diagnosis of gastric stromal tumors: a randomized crossover study. Endoscopy 2010;42:292ŌĆō299. 20354939.
14. Hoda KM, Rodriguez SA, Faigel DO. EUS-guided sampling of suspected GI stromal tumors. Gastrointest Endosc 2009;69:1218ŌĆō1223. 19394006.
15. Polkowski M, Gerke W, Jarosz D, et al. Diagnostic yield and safety of endoscopic ultrasound-guided trucut [corrected] biopsy in patients with gastric submucosal tumors: a prospective study. Endoscopy 2009;41:329ŌĆō334. 19340737.
16. Polkowski M, Larghi A, Weynand B, et al. Learning, techniques, and complications of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Technical Guideline. Endoscopy 2012;44:190ŌĆō206. 22180307.
17. Fazel A, Draganov P. Interventional endoscopic ultrasound in pancreatic disease. Curr Gastroenterol Rep 2004;6:104ŌĆō110. 15191687.
18. Dumonceau JM, Polkowski M, Larghi A, et al. Indications, results, and clinical impact of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 2011;43:897ŌĆō912. 21842456.
19. Moon JS. Endoscopic ultrasound-guided fine needle aspiration in submucosal lesion. Clin Endosc 2012;45:117ŌĆō123. 22866250.
20. Lee JH, Choi KD, Kim MY, et al. Clinical impact of EUS-guided Trucut biopsy results on decision making for patients with gastric subepithelial tumors >/= 2 cm in diameter. Gastrointest Endosc 2011;74:1010ŌĆō1018. 21889136.
21. Song HJ, Park YS, Seo DW, et al. Diagnosis of mediastinal tuberculosis by using EUS-guided needle sampling in a geographic region with an intermediate tuberculosis burden. Gastrointest Endosc 2010;71:1307ŌĆō1313. 20417504.
22. Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol 2002;33:459ŌĆō465. 12094370.
23. Dumonceau JM, Koessler T, van Hooft JE, Fockens P. Endoscopic ultrasonography-guided fine needle aspiration: relatively low sensitivity in the endosonographer population. World J Gastroenterol 2012;18:2357ŌĆō2363. 22654426.
24. Erickson RA, Sayage-Rabie L, Beissner RS. Factors predicting the number of EUS-guided fine-needle passes for diagnosis of pancreatic malignancies. Gastrointest Endosc 2000;51:184ŌĆō190. 10650262.
25. Jhala NC, Jhala DN, Chhieng DC, Eloubeidi MA, Eltoum IA. Endoscopic ultrasound-guided fine-needle aspiration. A cytopathologist's perspective. Am J Clin Pathol 2003;120:351ŌĆō367. 14502798.
26. Polkowski M. Endoscopic ultrasound and endoscopic ultrasound-guided fine-needle biopsy for the diagnosis of malignant submucosal tumors. Endoscopy 2005;37:635ŌĆō645. 16010608.
27. Mekky MA, Yamao K, Sawaki A, et al. Diagnostic utility of EUS-guided FNA in patients with gastric submucosal tumors. Gastrointest Endosc 2010;71:913ŌĆō919. 20226456.
28. Thomas T, Kaye PV, Ragunath K, Aithal G. Efficacy, safety, and predictive factors for a positive yield of EUS-guided Trucut biopsy: a large tertiary referral center experience. Am J Gastroenterol 2009;104:584ŌĆō591. 19262518.
29. Storch I, Jorda M, Thurer R, et al. Advantage of EUS Trucut biopsy combined with fine-needle aspiration without immediate on-site cytopathologic examination. Gastrointest Endosc 2006;64:505ŌĆō511. 16996340.
30. Iglesias-Garcia J, Poley JW, Larghi A, et al. Feasibility and yield of a new EUS histology needle: results from a multicenter, pooled, cohort study. Gastrointest Endosc 2011;73:1189ŌĆō1196. 21420083.
31. Bang JY, Varadarajulu S. Procore and flexible 19 gauge needle can replace trucut biopsy needle? Clin Endosc 2013;46:503ŌĆō505. 24143312.
Fig.┬Ā1Study patients. EUS, endoscopic ultrasonography; SET, subepithelial tumor; G, gauge; FNA, fine needle aspiration; TCB, Trucut biopsy; GIST, gastrointestinal stromal tumor.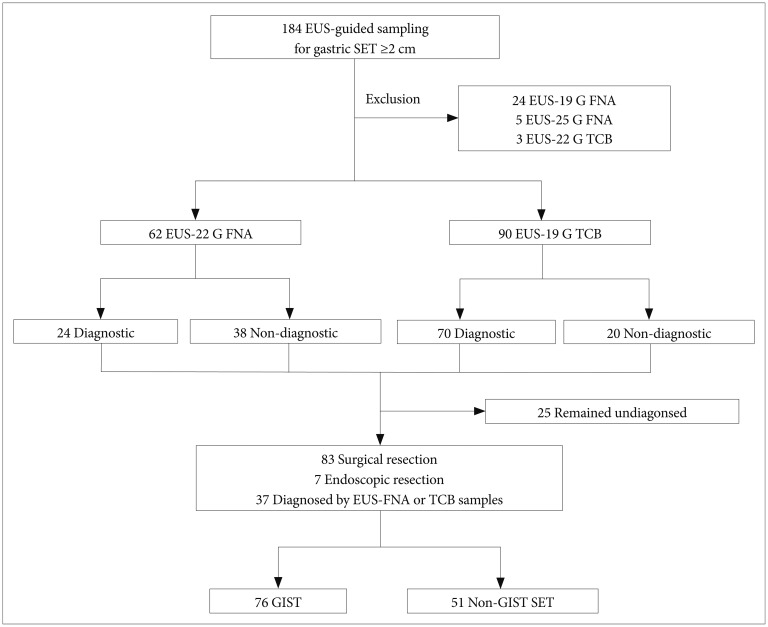
Fig.┬Ā2Diagnostic yields of 22-gauge (G) fine needle aspiration (FNA) and 19 G Trucut biopsy (TCB).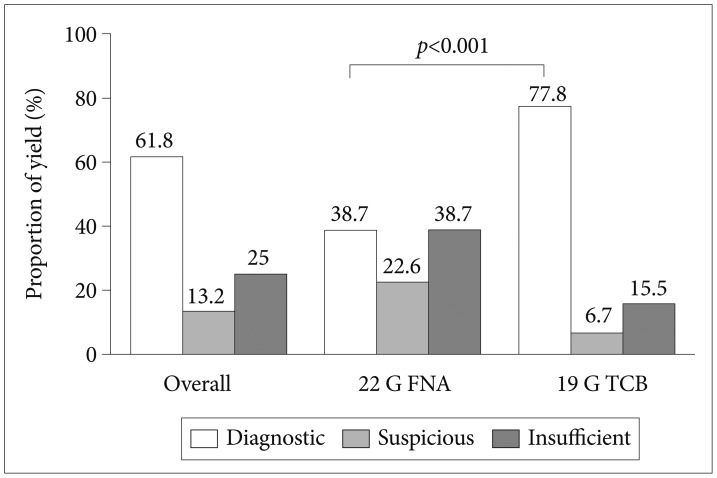
Fig.┬Ā3Diagnostic yields of gastrointestinal stromal tumors (GISTs) and non-GIST gastric subepithelial tumors. SET, subepithelial tumors; G, gauge; FNA, fine needle aspiration; TCB, Trucut biopsy.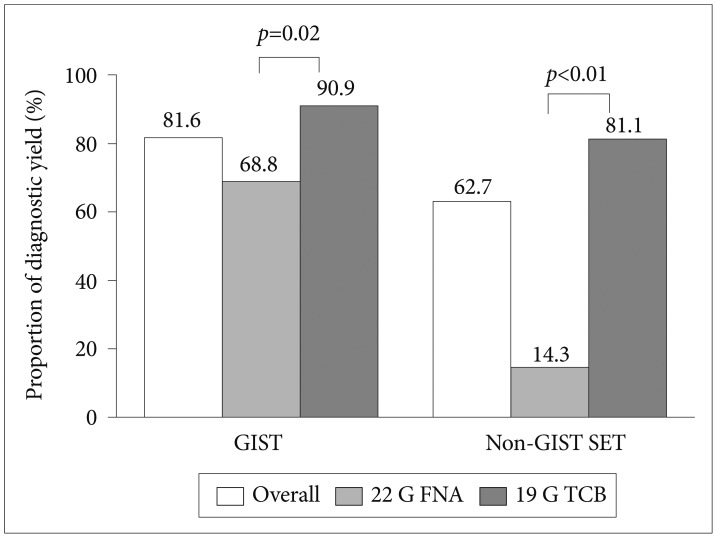
Table┬Ā1Final Pathology of Gastric Subepithelial Tumors Which Were Sampled by 22 G Fine Needle Aspiration and 19 G Trucut Biopsya)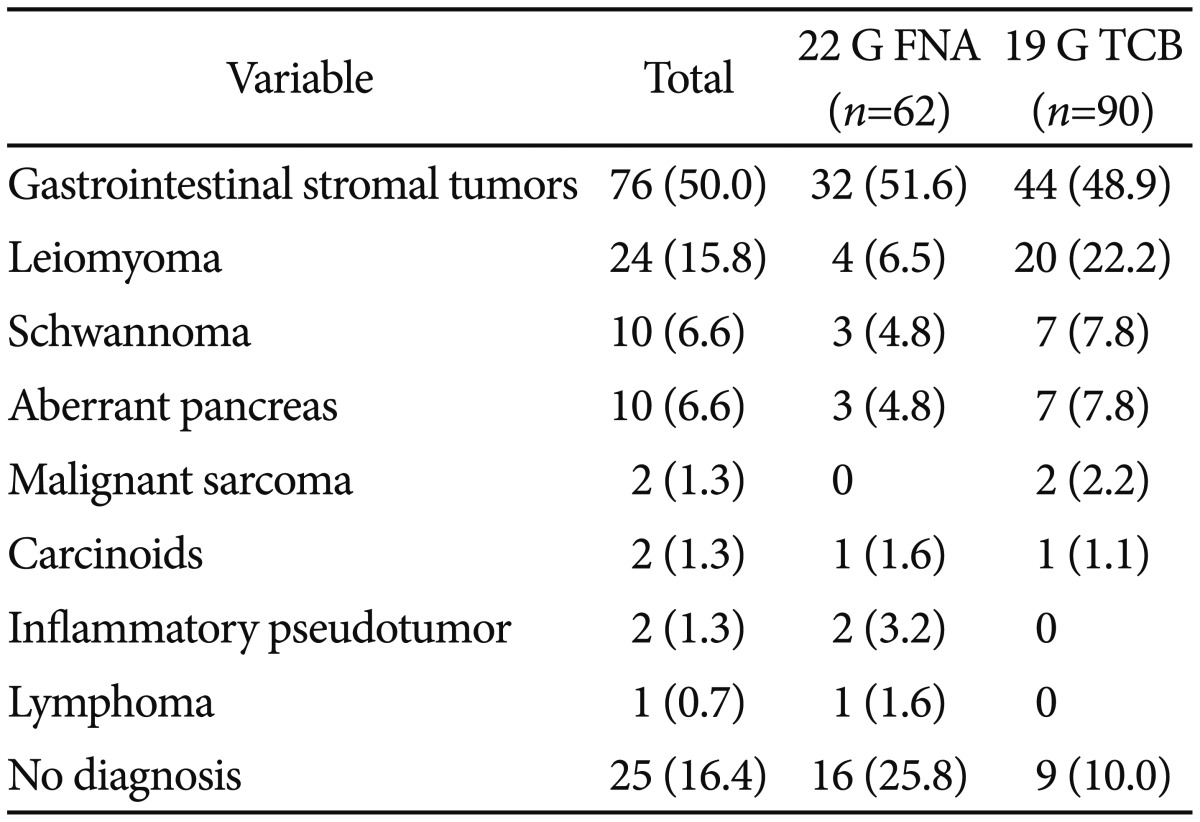
|
|
||||||||||||||||||||||||||||||||||||||||
