INTRODUCTION
Rhus-related illnesses in Korea are mostly caused by ingestion of the Rhus tree. It produces systemic contact dermatitis rather than allergic contact dermatitis.1 Some Koreans still enjoy eating the early shoots of the Rhus tree in the spring, which have been used as a folk remedy for functional gastrointestinal problems, musculoskeletal pain, and menstrual pain. Although a wide variety of mucocutaneous events, such as systemic contact dermatitis, have been reported to be elicited by the Rhus tree, cases of eosinophilic gastroenteritis (EGE) without involvement of the skin are rare. Here, we present a case of EGE caused by Rhus ingestion.
CASE REPORT
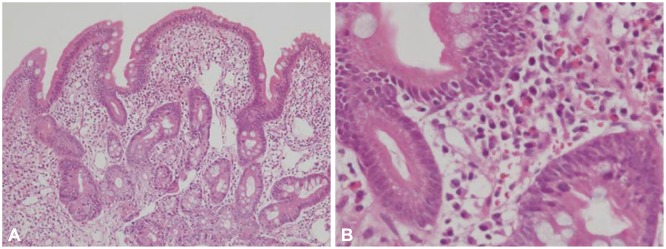
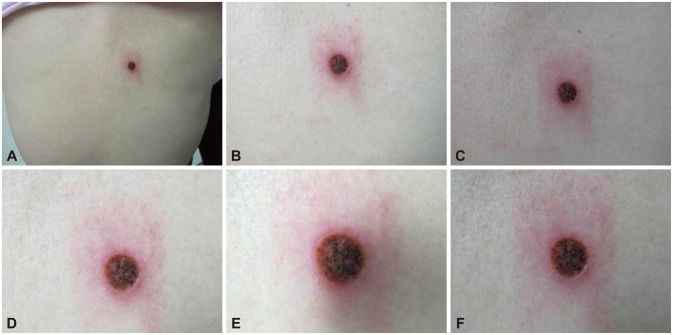
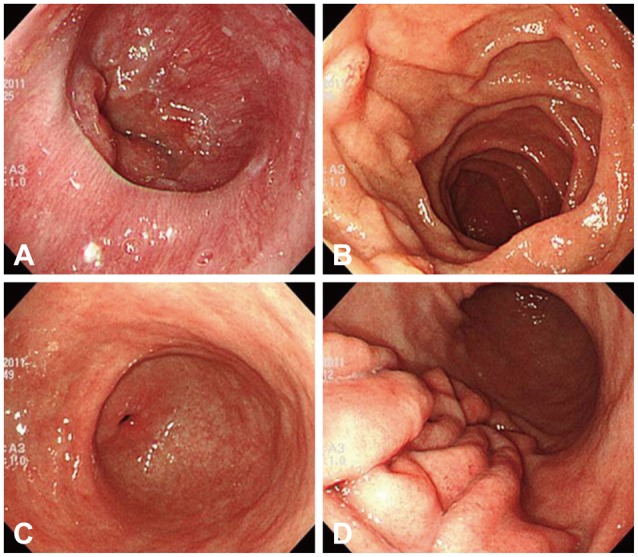
A 75-year-old woman was admitted to the emergency room complaining of hematemesis and hematochezia. She also had epigastric pain, nausea, and vomiting but no fever, weight loss, or rash. Her blood pressure was 110/70 mm Hg, heart rate 72 beats per minute, and respiratory rate 18 breaths per minute. She ingested Rhus extract 2 days prior. Laboratory examination results were as follows: leukocytes, 20,900/mm3 (neutrophils, 82%; lymphocytes, 8.9%; eosinophils, 0.1%; monocytes, 8.9%; basophils, 0.1%); hemoglobin, 14.4 g/dL; platelets, 86,000/mm3; and immunoglobulin E, 81.90 IU/mL (normal range, 0 to 100). The albumin level was low (2.9 g/dL; normal range, 3.5 to 5.5 g/dL) but other liver and renal function indicators were within the normal range. Acute phase reactants were elevated (C-reactive protein, 13.8 mg/dL; erythrocyte sedimentation rate, 22 mm/hr). Parasitologic examination and bacterial culture of stool results were normal. Abdominal computed tomography revealed edematous wall thickening of the duodenum and proximal jejunal loops (Fig. 1). Endoscopy showed friable and granular mucosal changes with touch bleeding in the second portion of the duodenum (Fig. 2). During the endoscopic examina-tion, multiple duodenal biopsies were taken. Pathologic find-ings showed edematous gastric mucosa and diffusely infiltrated inflammatory cells containing over 100 eosinophils per high power field, consistent with EGE (Fig. 3). For the evaluation of allergy, we performed a patch test with Rhus extracts ingested by the patient. We applied the Rhus extract-containing patch to the patient's back for 48 hours and then removed it. Seventy-two hours after patch application, the skin showed erythematous infiltration with ulceration, indicating a strong positive reaction (Fig. 4). We concluded that the Rhus extract provoked a type IV hypersensitivity reaction on the skin, and that it may be the allergen causing EGE. After avoidance of the allergen, the patient's symptoms remitted. One-month follow-up endoscopy showed improving mucosal edema and nodularity in the duodenum (Fig. 5).
DISCUSSION
Rhus tree belongs to the family Anacardiaceae, which includes cashew nuts, pistachio nuts, hog plum, Jamaican plum, sumac, mango, and poison ivy. Contact dermatitis occurrence after ingestion of Rhus-related food is very common in Korea.2,3 However, literature review suggests that Rhus-related gastrointestinal disease is very rare.
The pathomechanism of Rhus-related skin lesions is as follows: the active allergenic component of the Rhus tree is pentadecylcatechol (PDC), which is found in urushiol. However, PDC is not a complete antigen. PDC acts as a hapten, which becomes a complete antigen after binding to certain carrier proteins in skin. Antigen presenting cells in the skin, Langerhans cells and keratinocytes, present the urushiol antigen to both CD8+ T cells and CD4+ T cells through an endogenous or exogenous (endosomal) antigen-processing pathway, respectively. The small number of memory cells that remain in the body after the sensitization will respond to the allergen, resulting in the development of symptoms.4 In this case, sensitization to the allergen might have occurred through the cutaneous route. After ingestion of the Rhus extract, it may be taken up by intestinal epithelial cells. Dendritic cells might then take up the antigens and initiate the immune reaction with other costimulatory cytokines.5
Upon systemic exposure to urushiol, a presensitized person produces Rhus-related symptoms after a latency period. Presensitization is regarded as essential for the development of contact dermatitis. However, a positive patch test can be elicited in only one-third of patients with systemic contact dermatitis caused by Rhus chicken. The present patient had a positive skin patch test result for Rhus, which means the patient was presensitized to urushiol. However, she denied a past history of exposure to Rhus tree extracts. In the past, traditional furniture painted with Rhus lacquer was very popular in Korea. Previous reports in Korea showed that about 10% to 30% of the general population had allergic sensitization to Rhus lacquer.1 Even if the patient denied prior exposure, presensitization to Rhus, which was proven by a skin patch test, might have had a role in the development of EGE causing small intestinal bleeding.
In this case, EGE involved only the small intestine. The small intestine is the most frequently involved organ in patients with EGE, according to recent studies.6,7 Because the small intestine has the most abundant lymphatic tissue in the alimentary tract, it is the most active immune reaction site.8 This may be the reason it is most frequently involved in EGE. Of note, the patient had no peripheral blood eosinophilia. Although patients with EGE of the small intestine had the highest proportion of peripheral blood eosinophilia in a recent Japanese study, other studies showed that eosinophilic gastrointestinal disease typically occurs without peripheral blood eosinophilia more than 50% of the time.9 It is possible that because the involved gastrointestinal tissue was not sufficiently large, peripheral eosinophilia was not present in this case.
There is no consensus on the optimal treatment of EGE. If there is an identified allergen, avoidance improves clinical symptoms and also decreases mucosal eosinophils in patients with EGE.10,11 Steroids are the primary drugs for EGE. Antihistamines, mast cell stabilizers, and leukotriene antagonists can also be used.11,12,13
We report an interesting case of EGE caused by Rhus without involvement of skin. Although there are several reports describing contact dermatitis associated with Rhus ingestion, this case of EGE is the first reported. It may be necessary to educate patients, especially those with a history of exposure, about the harmful effect of Rhus.