Predictors of Rebleeding in Upper Gastrointestinal Dieulafoy Lesions
Article information
Abstract
Background/Aims
Dieulafoy lesions (DLs) are a rare but significant cause of upper gastrointestinal bleeding. We aimed to define the clinical significance of rebleeding and identify the predictors of rebleeding and mortality in upper gastrointestinal Dieulafoy lesions (UGIDLs).
Methods
Patients diagnosed with UGIDLs between January 2004 and June 2013 were retrospectively evaluated. Multivariate logistic regression analyses were performed to define the predictors of rebleeding and mortality in patients with UGIDLs.
Results
The study group consisted of 81 male and 36 female patients. Primary hemostasis was achieved in 115 out of 117 patients (98.3%) with various endoscopic therapies. Rebleeding occurred in 10 patients (8.5%). The mortality rate was significantly higher in patients with rebleeding than in those without rebleeding (30.0% vs. 4.7%, p=0.020). Multivariate logistic regression analysis revealed that kidney disease (p=0.006) and infection (p=0.005) were significant predictors of rebleeding in UGIDLs and that kidney disease (p=0.004) and platelet count (p=0.013) were significant predictors of mortality.
Conclusions
Rebleeding has an important prognostic significance in patients with UGIDLs. Kidney disease and infection are major predictors of rebleeding and mortality in patients with UGIDLs.
INTRODUCTION
Dieulafoy lesions (DLs) are a rare but important cause of upper gastrointestinal bleeding (UGIB). Advances in endoscopy have increased the rate of DL detection and provided novel and effective therapeutic approaches.1 The rate of primary hemostasis with endoscopic treatment can reach 75% to 100%.2 The evolution of endoscopic methods of hemostasis has markedly reduced the need for surgery in the management of DLs and significantly decreased the mortality rate from 80% to approximately 10%.13 However, DLs remain a predictor of unfavorable outcome in nonvariceal UGIB4 and is associated with frequently recurrent UGIB, which occurs in spite of repeated endoscopic therapy.5 The reported risk of rebleeding due to DLs is between 9% and 40%.1 Although the potential for rebleeding is high, the clinical significance of rebleeding DLs has not yet been determined. Furthermore, few studies have investigated the predictors of rebleeding and mortality in upper gastrointestinal Dieulafoy lesions (UGIDLs). Therefore, the objectives of our study were to define the clinical significance of rebleeding and identify the predictors of rebleeding and mortality in patients with UGIDLs.
MATERIALS AND METHODS
Patients
This was a retrospective study of the patients who were admitted to Chonnam National University Hospital with a diagnosis of UGIDL between January 2004 and June 2013. Data regarding the demographics, endoscopic findings, details of endoscopic therapy, recurrence of bleeding, and mortality due to UGIDLs were collected.
Endoscopic findings and treatment
During the study period, 7,877 patients with UGIB were admitted to our hospital. UGIDLs were endoscopically diagnosed in 206 of these patients. Two experienced endoscopists reviewed the endoscopic findings and excluded 87 patients whose endoscopic findings were more compatible with a small ulcer with an exposed vessel rather than UGIDLs. Two additional patients were excluded because they had another potential major bleeding focus (one ulcer with exposed vessel and one gastric angiodysplasia). Therefore, typical DL was diagnosed in 117 of the 7,877 patients (1.5%) (Fig. 1). The endoscopic visual criteria of DL were (1) active arterial spurting or micropulsatile streaming from a minute (<3 mm) mucosal defect or from normal surrounding mucosa; (2) visualization of a protruding vessel with or without active bleeding within a minute mucosal defect or normal surrounding mucosa; or (3) the appearance of a fresh, densely adherent clot with a narrow point of normal-appearing mucosa.6 We performed this retrospective study in accordance with the guidelines of the Institutional Review Board of Chonnam National University Hospital (CNUH-2014-012).

Flow diagram of the study showing entries and outcomes of all patients. UGIB, upper gastrointestinal bleeding; DLs, Dieulafoy lesions; UGIDLs, upper gastrointestinal Dieulafoy lesions; AKI, acute kidney injury; CKD, chronic kidney disease; DIC, disseminated intravascular coagulation; TAE, transcatheter arterial embolization.
Upper endoscopy was carried out within 24 hours of admission. The endoscopic therapeutic options for DL were epinephrine (1:10,000) injection, hemoclipping, band ligation, argon plasma coagulation, or combination treatment.
Definitions
Rebleeding was defined as one or more of the ongoing bleeding signs, including fresh hematemesis, hematochezia, fresh blood aspirated via a nasogastric tube, instability of vital signs, or a reduction of hemoglobin by more than 2 g/dL after 12 hours of primary hemostasis.7 When recurrent bleeding was suspected, endoscopic retreatment was carried out immediately. If bleeding was not controlled using the above-mentioned endoscopic procedures, surgical or interventional radiologic management was attempted. Long-term follow-up over 6 months was performed in all patients.
We defined kidney disease as either an acute kidney injury or chronic kidney disease based on KDIGO (Kidney Disease: Improving Global Outcomes) criteria. Acute kidney injury was defined by the presence of any of the following: increase in serum creatinine by ≥0.3 mg/dL (≥26.5 µM/L) within 48 hours; increase in serum creatinine by ≥1.5 times baseline, which is known or presumed to have occurred within 7 days; or urine volume <0.5 mL/kg/hr for 6 hours.8 Chronic kidney disease was defined as abnormalities of kidney structure or function, present for >3 months, with health implications.9 Infection was defined as a multiplication of microbes on the surface or in the lumen of the host causing signs and symptoms of illness or disease.10
Statistical analysis
The chi-square test and Mann-Whitney test were used to evaluate baseline characteristics. Categorical variables were analyzed by the chi-square test, while continuous variables were assessed by the Mann-Whitney test. Univariate and multivariate logistic regression analyses were used to detect independent predictors related to rebleeding and mortality during the follow-up periods. A p<0.05 was considered significant for all tests. Analyses were performed using SPSS version 20.0 (IBM Co., Armonk, NY, USA).
RESULTS
Clinical characteristics of the patients with UGIDLs
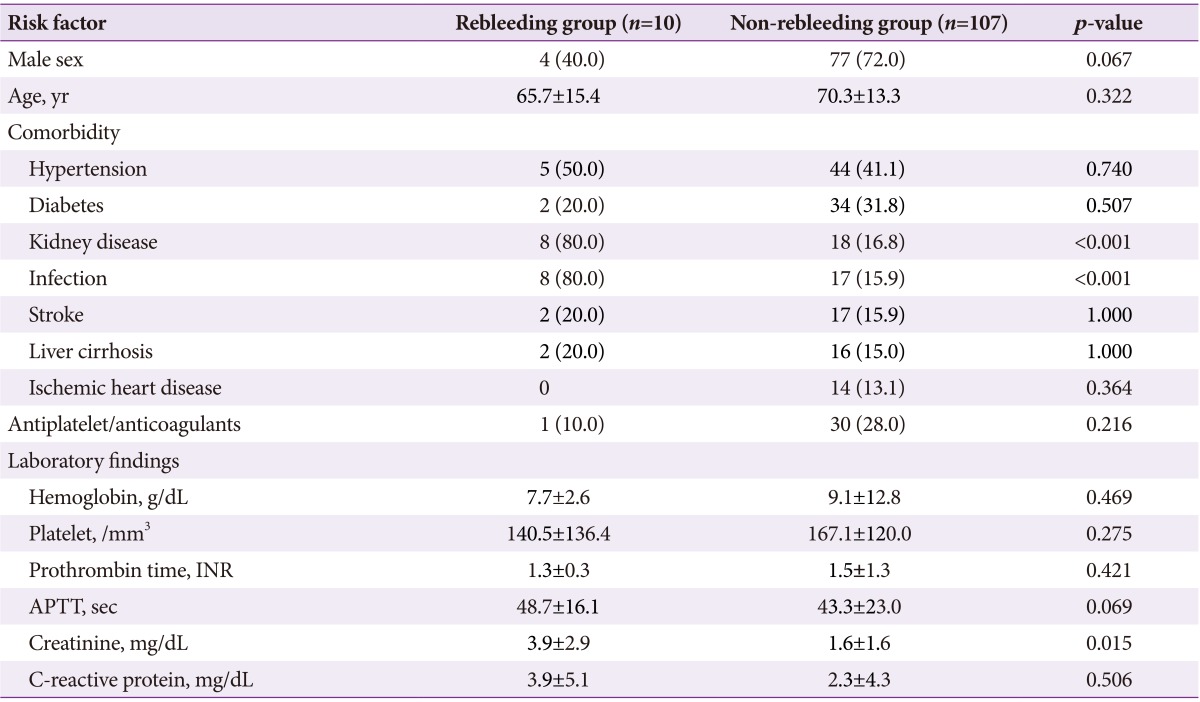
The UGIDL group consisted of 81 male and 36 female patients with a mean age of 69.9±13.4 years. Most patients had comorbidities such as hypertension (41.8%), diabetes (30.7%), kidney disease (22.2%), infection (21.3%), stroke (16.2%), liver cirrhosis (15.3%), or ischemic heart disease (11.9%). Thirty-one patients (26.4%) took antiplatelet and anticoagulants. Rebleeding occurred in 10 patients (8.5%), with a mean duration of 2.8 days after primary hemostasis. Differences in demographic data and clinical characteristics of the patients in the rebleeding group and non-rebleeding group are summarized in Table 1. Kidney disease and infection were significantly higher in the rebleeding group than in the non-rebleeding group. Serum creatinine levels were also significantly higher in the rebleeding group. In the 10 patients with rebleeding UGIDLs, six had one episode of rebleeding (1, 1, 1, 1, 5, and 6 days, respectively, after primary hemostasis), three patients had two episodes (3 and 5 days, 3 and 9 days, and 3 and 56 days, respectively, after primary hemostasis), and one patient had three episodes (4, 6, and 12 days, respectively, after primary hemostasis).
Endoscopic findings and treatments
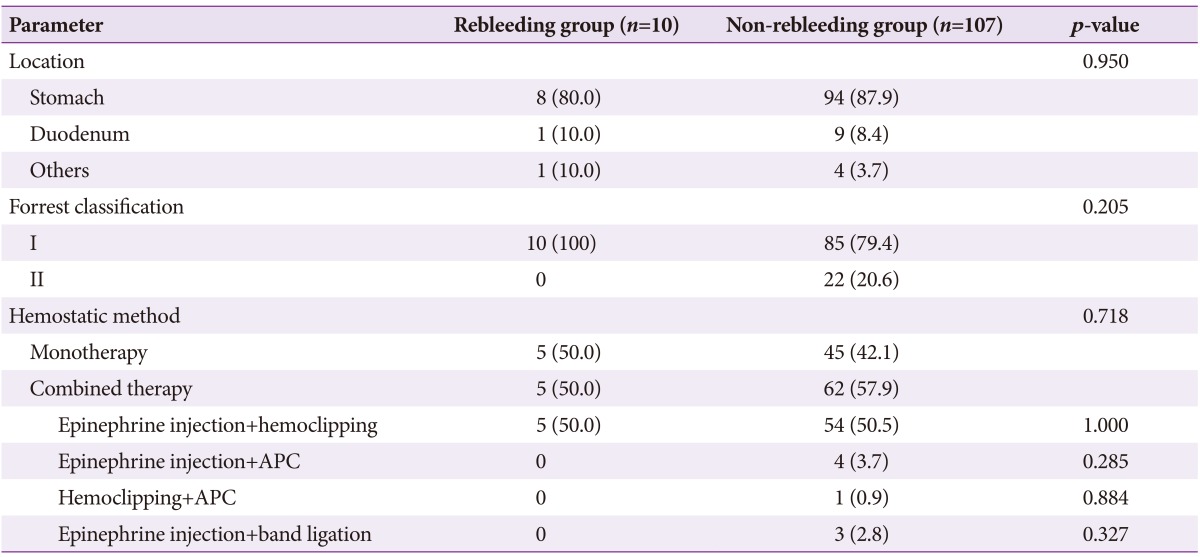
The most common location of UGIDLs was the stomach (87.2%). Duodenal bleeding was present in 8.5% of patients, and bleeding at the stoma occurred in 4.3%. The endoscopic characteristics and treatment modality of patients in the rebleeding and non-rebleeding groups are summarized in Table 2. There was no difference in the rebleeding rate according to the bleeding location. According to Forrest classification, oozing hemorrhage was the most common (61.5%), followed by spurting hemorrhage (19.7%) and Forrest type IIa to IIc (18.8%). No significant difference in the rebleeding rate was observed according to the Forrest classification. Primary hemostasis was successfully achieved by endoscopic therapy in 115 of patients with UGIDLs (98.3%; monotherapy in 42.1% and combination treatment in 57.9%). Epinephrine injection combined with hemoclipping was the most frequently used method (50.5%), followed by epinephrine injection with argon plasma coagulation (3.7%), hemoclipping with argon plasma coagulation (0.9%), and epinephrine injection with band ligation (2.8%). No difference in rebleeding rates according to endoscopic treatment modality was observed. Hemostasis was not achieved with epinephrine injection and hemoclipping in two patients; one patient was saved by transcatheter arterial embolization, but the other died of infection after surgery.
Clinical significance of rebleeding in patients with UGIDLs
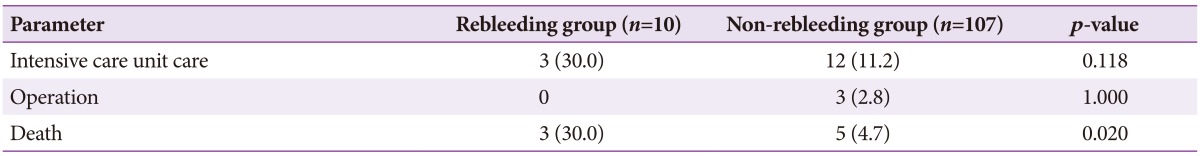
No significant differences were found in terms of intensive care unit care and operations received between groups. The overall mortality rate of patients with UGIDLs was 6.8% (8/117) in the current study and was significantly higher in the rebleeding group than in the non-rebleeding group (3/10 [30.0%] vs. 5/107 [4.7%], p=0.020) (Table 3). In the rebleeding group, three patients died of septic shock with acute kidney injury, whereas two patients had underlying chronic kidney disease. In the non-rebleeding group, deaths were due to acute kidney injury (two patients), infection with chronic kidney disease (one patient), infection (one patient), and disseminated intravascular coagulation due to colon cancer with multiple metastases (one patient) (Fig. 1). In total, six patients (6/8, 75%) died of kidney disease and five patients (5/8, 62.5%) died of infection; four patients (4/8, 50%) showed an overlap of both diseases.
Statistical analysis
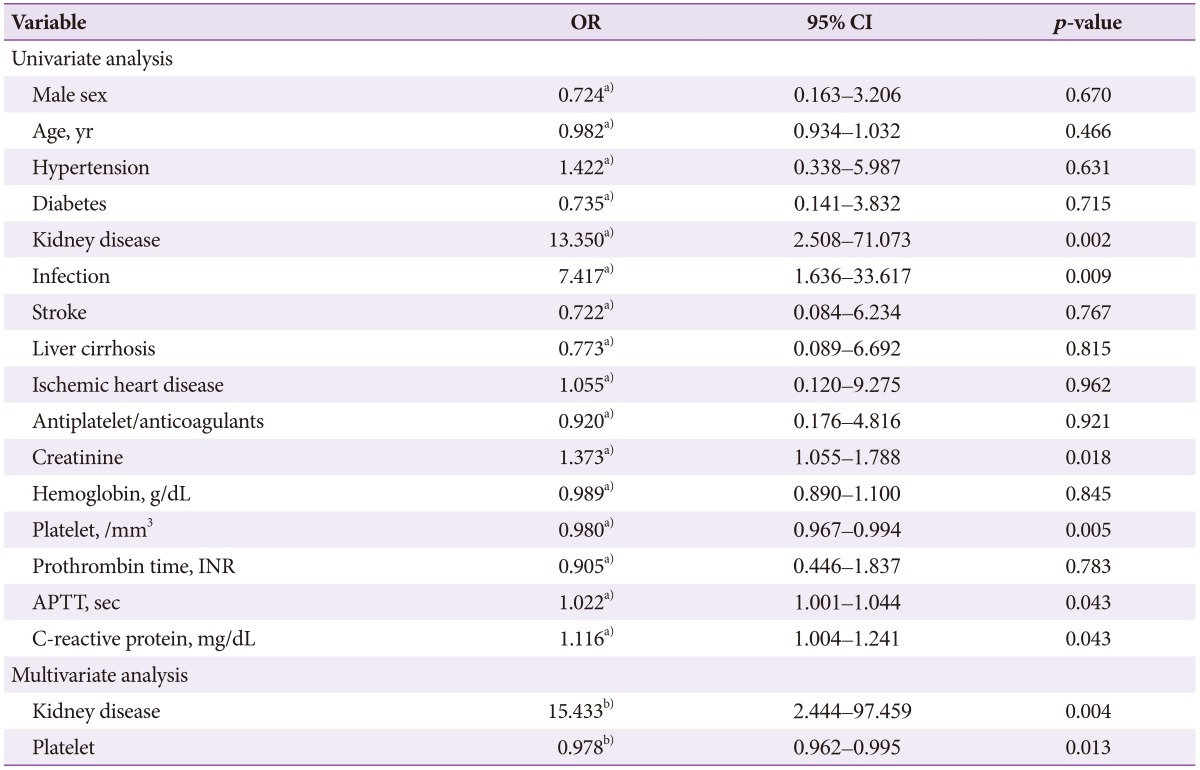
Results of univariate logistic regression analysis showed that male gender (conditional odds ratio [cOR], 0.20; 95% confidence interval [CI], 0.06 to 0.98; p<0.001), kidney disease (cOR, 19.77; 95% CI, 3.87 to 100.96; p<0.001), infection (cOR, 21.17; 95% CI, 4.13 to 108.50; p<0.001), and serum creatinine (cOR, 1.49; 95% CI, 1.16 to 1.91; p=0.001) were significant predictors of rebleeding in UGIDLs. Results of multivariate logistic regression analysis revealed that kidney disease (adjusted odds ratio [aOR], 11.35; 95% CI, 2.00 to 64.38; p=0.006) and infection (aOR, 12.32; 95% CI, 2.17 to 69.71; p=0.005) were significant predictors of rebleeding in UGIDLs (Table 4).
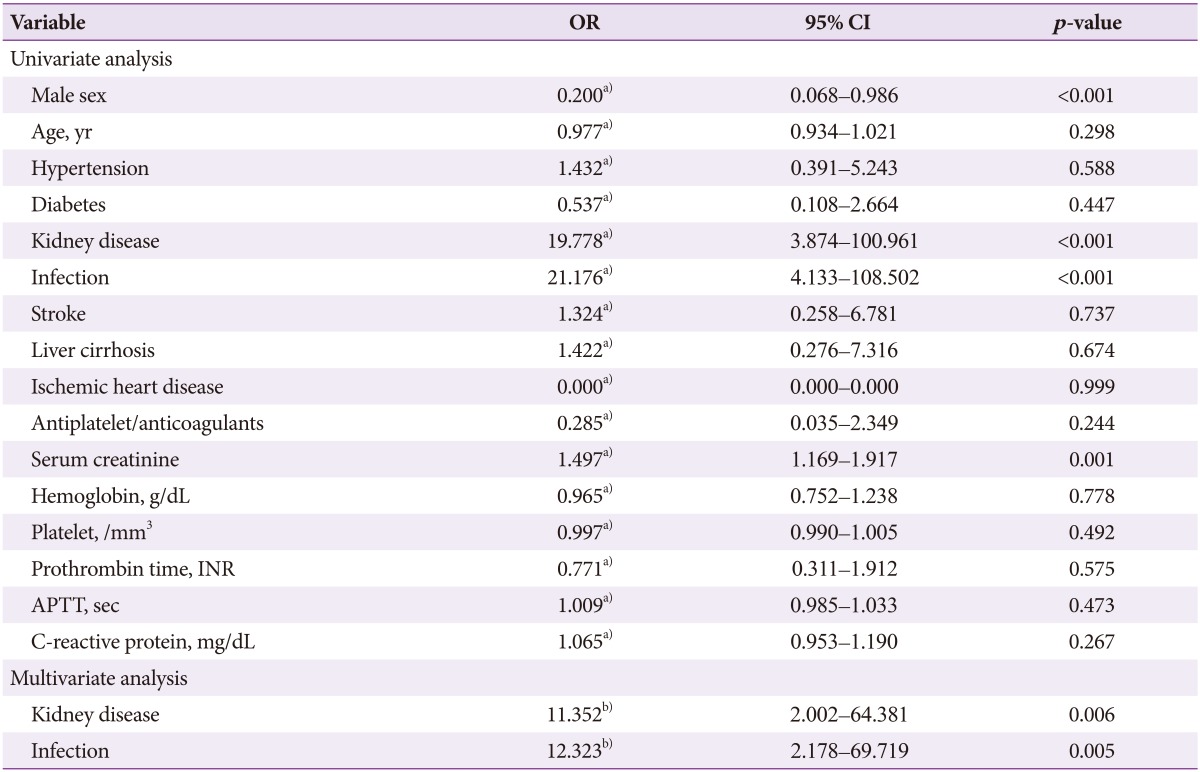
Univariate and Multivariate Logistic Analysis of the Independent Variables Related to Rebleeding in Upper Gastrointestinal Dieulafoy Lesions
According to univariate logistic regression analysis, significant predictors of mortality risk in patients with UGIDLs were kidney disease (cOR, 13.35; 95% CI, 2.50 to 71.07; p=0.002), infection (cOR, 7.41; 95% CI, 1.63 to 33.61; p=0.009), serum creatinine (cOR, 1.37; 95% CI, 1.05 to 1.78; p=0.018), platelet count (cOR, 0.98; 95% CI, 0.96 to 0.99; p=0.001), activated partial thromboplastin time (cOR, 1.02; 95% CI, 1.00 to 1.04; p=0.043), and C-reactive protein (cOR, 1.11; 95% CI, 1.00 to 1.24; p=0.043). In the multivariate logistic regression analysis, the significant predictors were kidney disease (aOR, 15.43; 95% CI, 2.44 to 97.45; p=0.004) and platelet count (aOR, 0.97; 95% CI, 0.96 to 0.99; p=0.013) (Table 5).
DISCUSSION
In the current study, the prevalence of UGIDLs was 1.5%. Primary hemostasis was successfully achieved by endoscopic therapy in 98.3% of patients with UGIDLs, and rebleeding occurred in 8.5% of patients. The mortality rate among patients with UGIDLs was 6.8% and was significantly higher in rebleeding patients than in non-rebleeding patients. Among patients with UGIDLs, kidney disease and infection were significant predictors of rebleeding, and kidney disease and platelet count were significant predictors of mortality.
The prevalence of UGIDLs in the current study is similar to that observed in other studies.11112 The incidence of DL has recently increased with the use of advanced endoscopic diagnostic techniques.13 This rise may also be explained by the increasing age of the population, which causes a subsequent increase in combined comorbidities. In addition, because DL is mainly diagnosed using endoscopic visual criteria, this rise may also be due in part to the misdiagnosis of small ulcers with an exposed vessel or the misdiagnosis of other vascular disorders as DL. In our study, after a thorough review by experienced endoscopists, 87 out of 204 cases (42.6%) that were initially classified as DL were later revealed to be other diseases, such as small ulcers with exposed vessels or other vascular disorders.
Many different types of endoscopic procedures were used to control bleeding in the current study, and a primary hemostasis rate of 98.3% was achieved. In recent years, several studies have shown that use of the hemoclip14 and endoscopic ligature improves the rates of primary hemostasis, reducing the need for subsequent endoscopy.1516 Theoretically, mechanical hemostasis leads to a smaller lesion of the surrounding tissue than thermal therapy or injection techniques, which suggests that these procedures could be a good first-line approach for the management of DL.171819 Another study observed better results for thermocoagulation vs. injection of a single substance.20 A recent study has concluded that combined therapy with hemoclip and Aethoxysklerol injection is the most effective,21 while other studies have shown that sclerosis with adrenaline was associated with a higher risk of bleeding recurrence.321 Similarly, Jamanca-Poma et al.22 reported that the recurrence of bleeding was associated with monotherapy with adrenaline and endoscopic findings of arterial spurting bleeding. In contrast to these other studies, we found no significant differences between patients in the rebleeding group and the non-rebleeding group in terms of the type of bleeding and the method of hemostasis used.
In the current study, the rate of rebleeding after primary hemostasis was 8.6%. According to the literature, the clinical significance of rebleeding in UGIDLs has not yet been determined. In the current study, rebleeding had an important prognostic significance in patients with UGIDLs. The mortality rate was significantly higher in patients with rebleeding than in those without rebleeding. Jamanca-Poma et al.22 reported that although many patients presented with comorbidity (mainly hypertension and ischemic heart disease), no significant relationship was found between comorbidity and rebleeding risk. In the current study, multivariate logistic regression analysis revealed that kidney disease and infection were significant predictors of rebleeding in patients with UGIDLs. In particular, rebleeding occurred at a higher rate when the combination of infection and kidney disease was not properly controlled by medical treatment.22 Therefore, more effective endoscopic management and control of underlying diseases are warranted to better prevent rebleeding in patients with UGIDLs. Our study included many patients with various comorbidities, such as hypertension, diabetes, kidney disease, infection, stroke, liver cirrhosis, and ischemic heart disease. Many other prospective studies of UGIDLs have excluded patients with such serious comorbidities. Thus, it was difficult to compare the current results with those of previous studies.215161718 To confirm our results, larger prospective studies are needed.
Advances in endoscopy have significantly decreased mortality rates among patients with UGIDLs (from 80% to 8.6%).1 The mortality rate observed in the current study was 6.8%. Improvements in prognosis might be explained by early endoscopy, aggressive endoscopic treatment, and careful assessment of the patient's overall health status.4 Although rebleeding frequently occurred in patients with bleeding despite repeated endoscopic therapy, no bleeding-related deaths in which the endoscopic hemostasis was effective occurred. Instead, the main causes of death were kidney disease and infection. According to the multivariate logistic regression analysis, kidney disease and low platelet count were significant predictors of mortality in patients with UGIDLs. Low platelet counts are a reflection of septic conditions. Therefore, kidney disease and infection were the major predictors affecting not only rebleeding but also mortality. The current results suggest that UGIDLs should be considered as a warning sign indicating deterioration of the underlying comorbid condition. Effective control of underlying diseases is essential to decrease the rebleeding rate and improve the prognosis of patients with UGIDLs. However, because few studies have demonstrated a relationship between comorbidity and rebleeding in patients with UGIDLs, larger prospective multicenter studies are needed.
The strengths of this study are that it included many patients with various comorbidities, and the number of enrolled patients was relatively large compared with previous other studies of DL.
Our study has some limitations. First, it is a retrospective, single-center study. To confirm our results, larger prospective multicenter studies are needed. Second, many types of endoscopic treatment were used. Thus, it was difficult to compare the therapeutic results among the specific endoscopic treatment modalities. However, a high success rate of primary hemostasis (98.3%) was successfully achieved in patients with UGIDLs, regardless of the endoscopic method used.
In conclusion, rebleeding has an important prognostic significance in patients with UGIDLs. Because kidney disease and infection are major predictors of rebleeding and mortality in patients with UGIDLs, control of the underlying disease is an important step to improve prognosis.
Notes
Conflicts of Interest: The authors have no financial conflicts of interest.