INTRODUCTION
Obesity has emerged as a major health threat in Korea, as well as globally, in recent years. According to Korea Health Statistics 2011, obesity affects one-third of Korean adults [1]. The importance of obesity should be emphasized because it is a major risk factor for cardiovascular disease, hypertension, and diabetes mellitus [2]. Obesity also increases the risk of comorbidities, such as gastroesophageal reflux disease (GERD), biliary disease, non-alcoholic fatty liver disease, non-alcoholic steatohepatitis, and gastrointestinal (GI) cancer [3]. However, the majority of obese patients fail to achieve sustained weight reduction or optimal weight by reduced calorie intake, a low-fat diet, exercise, or some combination thereof. If weight loss is achieved, it is usually difficult to reduce more than 10% of total body weight and sustain the effects for a prolonged period.
Bariatric surgery has shown good results in achieving adequate and reliable weight loss using either a laparoscopic approach or an open technique. The number of bariatric surgeries performed in the US has continued to increase annually, from 158,000 procedures in 2011 to 193,000 procedures in 2014 [4].
Bariatric surgery is not a straightforward procedure, although it has known benefits, such as long-term weight loss and improved obesity-related comorbidities. This is in part because it may result in complications that can be life threatening. Furthermore, postoperative GI and surgical complications can occur with different types of surgery, including anastomotic leaks, fistulas, stricture, GI bleeding, staple line disruption, bezoars, internal hernia, peritonitis, and band slippage, or migration [5]. Risk stratification may prevent many of these patients from gaining access to surgical interventions for weight loss. Endoscopic approaches are similar to surgical methods, although they are less invasive, safer, and more cost-effective than current surgical approaches. However, the evaluation of endoscopic bariatric procedures is limited by the small number of published studies and sample size, and the long-term effects are currently not well known. Finally, endoscopic restriction procedures have not shown additional remarkable benefits beyond surgery until now.
Thus, this article reviewed studies describing endoscopic gastroplasty and discussed its role in the management of obesity.
GASTROPLASTY AND SURGICAL GASTROPLASTY
Gastroplasty is a type of weight loss surgery to treat morbid obesity. It is a restrictive procedure in that it reduces the gastric cavity and limits the amount of food intake, whereas alternative procedures, such as gastric bypass, work by reducing the absorption of nutrients and calories in the digestive tract to limit caloric intake. Following gastroplasty, satiety can be achieved following the intake of a small amount of food due to a reduction in the size of the stomach cavity, thereby facilitating reduction in body weight. Additionally, gastroplasty does not cause nutritional deficiencies, including anemia, which occurs following gastric bypass because it does not involve alterations in the anatomy of the GI tract. However, the ingestion of high-caloric solid foods and fluids eventually occurs in most of these patients, which results in weight gain and recurrence of the associated comorbid conditions [6]. Strict follow-up and regular monitoring with well-planned dietary intake and exercise are beneficial to maintaining weight loss after gastroplasty.
In recent decades, various bariatric procedures have been performed, including vertical-banded gastroplasty (VBG), Roux-en-Y gastric bypass, biliopancreatic diversion, laparoscopic-adjustable gastric banding, and laparoscopic sleeve gastrectomy. VBG [7] and silastic ring vertical gastroplasty (SRVG) [8] are widely used gastroplasty procedures for managing obesity.
These procedures are performed by stapling the stomach in continuity and vertically, thereby leaving a 10ŌĆō30 cc upper proximal stomach pouch. A small gap in the staple-line is left, which causes digested food in the pouch to slowly descend into the distal stomach. To reduce the possibility of preventing stretching and enlarging of the passage into the distal stomach, a tight plastic band or a silastic ring is placed around the upper stomach.
Currently, most surgeons who perform gastroplasties transect the stomach to decrease the incidence of staple-line breakdown [6]. Laparoscopically, transection of the stomach is currently performed relatively easily when attempting various gastroplasties [9]. The outcome of gastroplasties in prospective clinical evaluations yields an average percentage excess weight loss of approximately 50% in approximately 50% of patients [10]. The advantages of gastroplasty operations include its simplicity, lack of a need to rerout the GI tract, reduced invasiveness, and no malabsorption. However, complications include vomiting, GERD, staple-line breakdown, band erosion, outlet stenosis, and obstruction. This is in addition to complications that are common to all other GI operations, such as wound infection, bleeding, incisional hernias, leaks, fistula formation, deep vein thrombosis, and pulmonary embolism [6].
ENDOSCOPIC BARIATRIC PROCEDURES AND ROLE
Recent surgical trends have sought to achieve a smaller scar and use a minimally invasive approach. Endoluminal bariatric procedures, which are performed using flexible endoscopy completely through the GI tract, offer the potential for an ambulatory weight loss procedure that may be more cost-effective and safer than current surgical bariatric procedures [11]. Endoscopic bariatric procedures have been attempted as a type of relatively noninvasive approach related to the restriction of food intake or malabsorption.
Several roles for endoscopy in the management of obesity have been suggested. For example, endoscopy can be used as an early intervention in patients with early stage obesity as a bridge to surgical procedures to reduce obesity-related operative risks; as a metabolic procedure that primarily addresses comorbid illnesses such as diabetes; as a primary obesity procedure that seeks outcomes similar to those of traditional bariatric surgery with reduced periprocedural risk; and, finally, as a revision procedure for failed bariatric surgical procedures [12]. However, the evaluation of endoluminal endoscopic bariatric procedures is limited by the small number of patients enrolled and the short follow-up period therein.
TYPES OF ENDOSCOPIC GASTROPLASTY
Transoral gastroplasty (with the TOGA system)
The Transoral Gastroplasty (TOGA) system (Satiety Inc., Palo Alto, CA, USA) represents the first endoscopic device designed primarily to create a vertical gastric sleeve. The TOGA system has two disposable staplers: the TOGA Sleeve stapler and the TOGA Restrictor. The TOGA Sleeve Stapler is introduced over the guide wire after dilation with the Savary bougie. A smaller gastroscope is advanced through the device and retroflexed to directly visualize the stapler. The sleeve stapler is tightened to acquire a double-fold of tissue with a strong vacuum, and the staples are then fired. This process is repeated to add a second staple line. Subsequently, the sleeve outlet is constricted using the TOGA restrictor, which is acquired with a vacuum and the stapler. Additional restrictions are in place to reduce the sleeve outlet from approximately 20 to 10 mm in diameter. This allows completion of a restricted gastroplasty along the lesser curvature of the proximal stomach [13]. Following the procedure, the amount of food or liquids that the patient can eat is limited by the size and anatomy of the gastric pouch, with an accompanying feeling of early satiety [14].
The first human multicenter study using the TOGA system has been reported. Twenty patients with a mean body mass index (BMI) of 43.3 (range, 35ŌĆō53) kg/m2 were enrolled. Although device-related adverse events, such as vomiting, pain, nausea, and transient dysphagia, were reported, no serious adverse events occurred. The mean weight loss of patients was 16.2% at 1 month, 22.6% at 3 months, and 24.4% at 6 months. However, a concern with this pilot study was the high incidence (61.9%; 13/21 patients) of gaps between the two staple lines at 6-months follow-up endoscopy [13].
The technique was subsequently improved by the development of an adjustable septum, thereby allowing closer apposition of the two staple lines to reduce the gap between the staple lines and the progression of the ischemic process that is related to transmural stapling. In a second human pilot study, there were no serious adverse events, and the mean weight loss was 19.2%, 33.7%, and 46.0% at 1, 3, and 6 months, respectively. The occurrence and size of the gap between the staple lines was reduced, but 36.3% (4/11) of the patients had a mid-stoma (<1 cm) visible endoscopically at 6 months [15].
Endoluminal vertical gastroplasty
The EndoCinchŌäó suturing system (C.R. Bard Inc., Murray Hill, NJ, USA) was originally designed for the endoluminal endoscopic treatment of GERD [16]. This system includes a capsule that is attached at the tip of the endoscope, a knot pusher, a tag pusher, tags attached to sutures, and a suture cutter [17]. Sutures were made by deploying the T-tag suture after sucking the mucosa and submucosa of the gastric wall into the capsule.
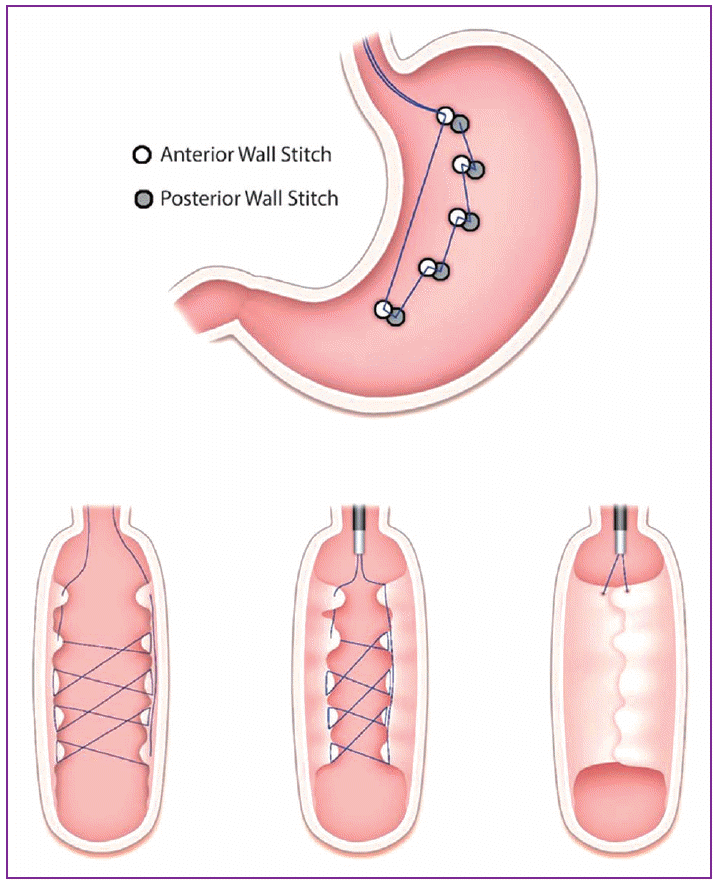
The first stitch is placed proximally in the nearest folds on the anterior face of the gastric fundus. Then, the stitch configuration comprises one continuous suture running through five to seven bites of tissue or stitch points. An illustration of a continuous stitch configuration is given in Fig. 1. After the stitches are placed and visualized, the suture is tightened, thereby bringing the anterior and posterior faces together to create the endoluminal vertical gastroplasty [18].
Fogel et al. [18] evaluated the feasibility and effectiveness of the EndoCinch suture system for weight loss in 64 patients (mean age=31.5 years). The mean procedure time was approximately 45 minutes. The patients experienced a significant reduction in BMI at 12 months (mean [SD] BMI=39.9┬▒5.1 kg/m2 vs. 30.6┬▒4.7 kg/m2, p<0.001) and the amount of weight loss, relative to total weight, was 21%, 39.6%, and 58.1%, at 1, 3, and 12 months after the procedure, respectively. However, there was some concern regarding the durability of the plication, as only approximately 35% of the suture configurations remained fully intact during the follow-up period.
The Restore Suturing System (Bard Davol, Warwick, RI, USA) is a modified version of the previous EndoCinch Suturing System that overcomes the requirement for an overtube, multiple esophageal intubations, and a second endoscope to position the fastening device. This device allows multistitch suturing and suture fastening to be performed during a single intubation.
A study using this device assessed the weight reduction of 18 patients (mean age=40 years) after 12 months. The average number of plications placed per patient was six, and the mean procedure time was 2.1 hours. At the 12-month follow-up, 14 patients achieved statistically significant decreases in mean weight (11.0 kg) and mean BMI (4.0 kg/m2) [19].
POSE
This procedure is performed using the Incisionless Operating Platform (IOP; USGI Medical, San Clemente, CA, USA). The device creates tissue plications by opposing tissue and then deploying and anchoring full-thickness stitches. It uses a four-channel platform with a visualization endoscope, a rotatable tissue grasper, and suture cutter (g-Prox), a tissue helix (g-Lix), and a suture anchor deployment catheter (g-Cath). During the primary obesity surgery endoluminal (POSE) procedure, stomach tissue is plicated in eight or nine locations in the fundus and in three or four locations in the distal body, using specialized suture anchors. The plicated areas mechanically and physiologically restrict contact between the ingested food and the full surface area of the stomach.
A recent study reported the results of the POSE procedure in 147 patients (mean age=43.8 years). The mean procedure time was approximately 35 minutes and an average of 7.5 and 3.5 suture anchors were placed in the fundus of the stomach and distal body, respectively. The baseline weight was significantly reduced at 3, 6, and 12 months after the POSE procedure (p<0.05). The patients experienced a mean total weight loss of 15 kg at 6 months, and the 116 patients who were available for follow-up at 1 year had a mean total weight loss of 16.6 kg (15.1%) and an excessive weight loss of 44.9% [20].
ENDOSCOPIC SLEEVE GASTROPLASTY
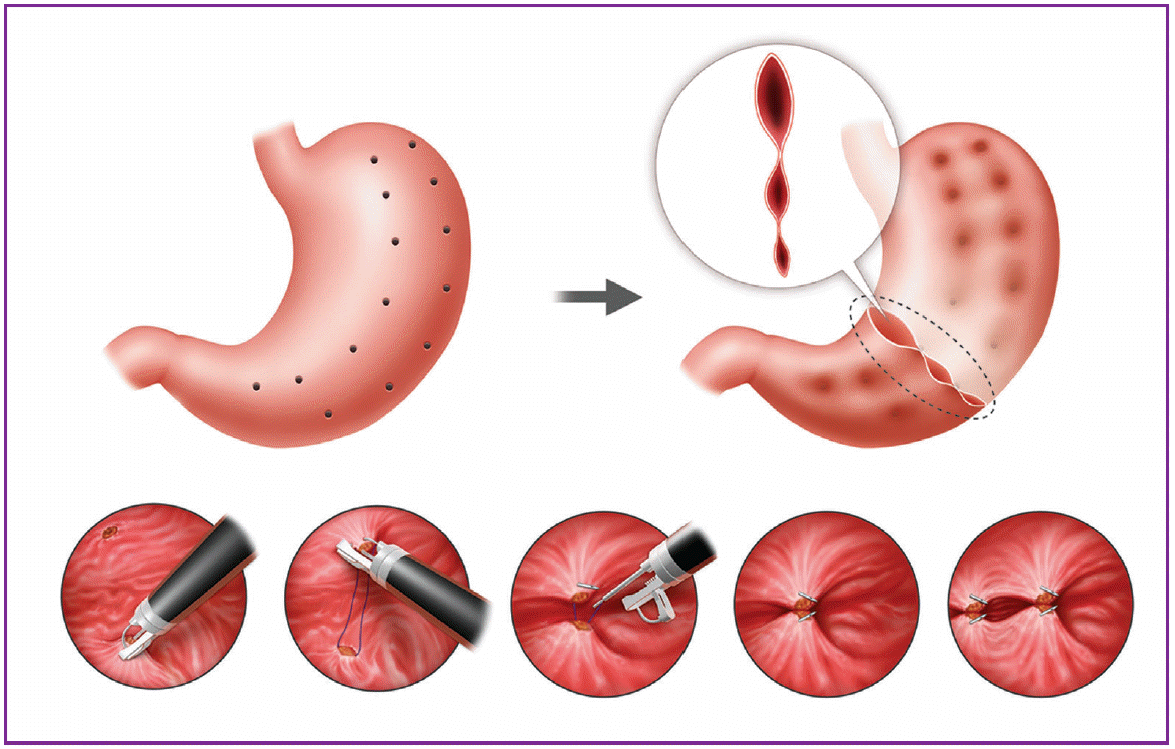
The endoscopic sleeve gastroplasty procedure is similar to a laparoscopic sleeve gastrectomy in that a portion of the stomach is isolated by creating an uninterrupted series of full-thickness sutures along the length of the stomach approximating the anterior, greater curvature, and posterior walls. An illustration of a continuous stitch configuration is given in Fig. 2. To reduce the endoluminal gastric volume, a cap-based flexible endoscopic suturing system (OverStitch; Apollo Endosurgery, Inc., Austin, TX, USA) is mounted onto a double-channel endoscope to achieve full-thickness. Then, sutures are run through the gastric wall from the antrum to the fundus [21].
A pilot study was performed to evaluate the feasibility of endoscopic sleeve gastroplasty in 20 patients with a mean age of 45.8 years. The initial body weight (108.5 kg) was significantly reduced by 8.2 kg (7.6%), 13.6 kg (12.4%), and 19.3 kg (17.8%), at 1, 3, and 6 months, respectively [22].
Recent experiences with endoscopic sleeve gastrectomy in 242 patients at three centers demonstrated that the percentage of total body weight loss was 16.8%, 18.2%, and 19.8%, at 6, 12, and 18 months, respectively. At the 18-month follow-up, 66.7% of patients sustained a greater than 15% total body weight loss [23].
CONCLUSION
Flexible endoscopic endoluminal approaches for obesity carry great potential, such as achieving good cosmetic effects via a scarless procedure. They are also safer, more cost-effective, do not require general anesthesia or intubation, require only short-term hospitalization, and involve less reconstruction compared with current surgical approaches. Despite these myriad positive possibilities, the evaluation of endoluminal procedures is limited by the small number of clinical studies and the short-term follow-up. In addition, most studies have been performed at only a few centers, with the special devices that are needed for bariatric endoluminal procedures not available in all countries. Further research should be conducted in different regions or countries since obesity rates differ by area. Furthermore, endoscopic gastroplasty should be carefully considered in areas with a high prevalence of Helicobacter pylori infection or gastric cancer due to limitations with respect to gastric cancer screening.
If the more vigorous clinical studies into endoscopic gastroplasty techniques currently in progress prove successful, these techniques are likely to become standard procedures for treating obesity in the near future.