INTRODUCTION
Over the last decade, esophagogastroduodenoscopy and colonoscopy have been widely acknowledged as optimal modalities for the examination and treatment of various gastrointestinal (GI) diseases because of their clinical and economic benefits compared with surgery. However, these procedures require much time and effort and include a significant risk for the patient. Especially difficult cases of endoscopic removal of foreign bodies and malignant lesions are very burdensome to both the patient and doctor [1]. Therefore, a new approach for these procedures is needed. However, current endoscopic platforms are limited by the narrow and complex GI tract. The outer diameters of a conventional endoscope are 1 cm in the gastroscope and 1.2 cm in the colonoscope. The endoscope channel is a tunnel space in which an external accessory tool can enter the GI tract. Conventional channel sizes are 2.8 mm for the gastroscope and 3.2 mm for the colonoscope. Endoscopic resection (e.g., endoscopic mucosal and submucosal resection) is a very common procedure and reduces the mortality from GI cancer [2-5]. However, many procedures are required in cases of multiple polyps larger than the endoscopic channel using conventional methods [6,7]. Such repeated insertions are difficult for the patient and operator and can cause musculoskeletal disorders in endoscopists. Several overtube devices have been designed to facilitate endoscopy [8-12]. However, the practical use of overtubes in hospitals is limited because of the incidence of various complications such as pharyngeal and esophageal perforation, variceal rupture, overtube separation, transient vocal cord paralysis, pneumomediastinum, and tracheal compression [9,13-18]. In addition, conventional simple overtubes are not used in Korea because they are expensive, except for esophageal drainage and variceal ligation. Hence, a new guidetube was developed to overcome these limitations. This study evaluated the feasibility of new endoscopic procedures using this guidetube.
MATERIALS AND METHODS
Participants
In July 2018, 5 expert endoscopists working at SMG-SNU Boramae Medical Center (Seoul, Korea) participated in this study. In each simulation, a timer was started when the scope was inserted into the entrance and stopped when the scope reached its target position.
Newly developed endoscopic guidetube
The new guidetube was designed to enhance secure insertion into the target area. It is composed of inner and outer tubes (Fig. 1). The outer tube is made of silicone while the inner tube is composed of a plastic channel. The outer tube has a 15-mm outer diameter and 13-mm inner diameter. To reduce mucosal trauma caused by rigidity of the tube, silicone was used for the tube material, as it will be inserted in the urethra and bladder, which are sensitive to trauma. The plastic channel allows the passage of various endoscopic instruments (biopsy forceps, snare, etc.). The basic guidetube is 30-cm long and is mainly used for the stomach and sigmoid colon. However, it can be manufactured or cut into various lengths by the manufacturer or endoscopist. Guidetubes of 80 cm and 60 cm in length can be used for the ascending and transverse colons.
Procedures using the guidetube in the stomach and colon simulator
We performed a series of endoscopic procedures with a gastroscope (GIF-260; Olympus, Tokyo, Japan) in 2 GI simulators: Endoscopy Gastrointestinal ERCP Upper Gi Training Simulator Deluxe-1 and Colonoscopy Training Simulator Manikin (Buyamag Inc., Carlsbad, CA, USA).
The guidetube was inserted and fixed at the target site of the stomach or colon under endoscopy guidance. Fixed sites included the sigmoid colon (anal verge [AV]: 30 cm), descending colon (AV: 50 cm), transverse colon (AV: 60 cm), ascending colon (AV: 70 cm), and esophagus (upper incisor teeth: 30 cm). Five expert endoscopists repeatedly inserted the guidetube at the above sites for 20 times. For more practical testing, large masses (large pork loaf, pen cover) were placed on the above points, and endoscopic retrieval using the guidetube was repeated 20 times.
A total of 15 endoscopic procedures were performed in each GI simulator (Fig. 2): 4 repeated endoscopic insertions (2 conventional and 2 guidetube-assisted) in a stomach model, 8 repeated endoscopic insertions (4 conventional and 4 guidetube-assisted) in a colorectal model, and retrieval of multiple foreign bodies (1 conventional and 1 guidetube-assisted), multiple polyps (1 conventional and 1 guidetube-assisted), and large amounts of food (1 conventional and 1 guidetube-assisted).
Statistical analysis
Statistical analysis was performed using the paired, one-sided StudentŌĆÖs t-test. A p-value <0.05 was considered to be statistically significant. Insertion time was calculated based on each stomach and colon position using the guidetube. The effectiveness of the endoscopic procedure using the guidetube was verified by comparing the results with standard endoscopic insertion at SMG-SNU Boramae Medical Center.
RESULTS
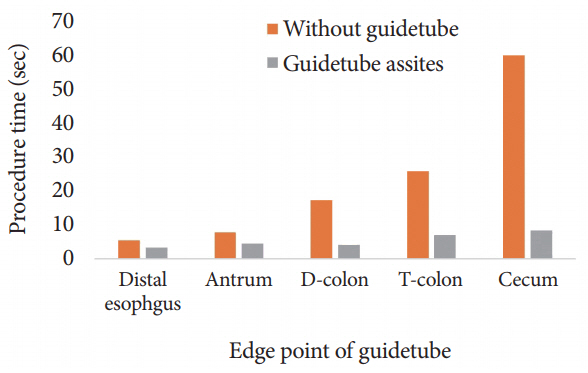
The primary outcome measure was the total procedure time. Endoscopic insertion times were reduced by approximately 70% using the guidetube. The mean insertion time of endoscope with the guidetube was significantly shorter than that without the guidetube: distal esophagus (3.2┬▒0.5 sec vs. 5.4┬▒1.8 sec); distal gastric antrum (4.4┬▒0.5 sec vs. 5.4┬▒1.8 sec); descending colon (4┬▒1 sec vs. 17.2┬▒1.9 sec); transverse colon (7┬▒1.2 sec vs. 25.8┬▒1.5 sec); and cecum (8.4┬▒0.89 sec vs. 60┬▒8.4 sec). Details are presented in Table 1 and Fig. 3.
The secondary outcome measure was safety and efficiency. The guidetube was safely inserted without scratch using low pushing force. Its pushing force was lower than the simple overtube (proximal esophagus: 1.5┬▒0.1 kg vs. 4.4┬▒0.1 kg). Objects of various sizes larger than the endoscopic channel were easily removed using the guidetube-assisted endoscopic procedures. The mean time to retrieve a 2-cm polyp with the guidetube was significantly shorter than that without a guidetube (descending colon: 15.2┬▒1.5 sec vs. 36┬▒3.1 sec).
DISCUSSION
Many endoscopic procedures (removal of multiple large polyps or sharp foreign bodies) are challenging and time-consuming. They require repeated endoscopic insertion, which causes much pain for the patient and repetitive motion injuries for the operator. Consequently, many overtubes have been developed to make endoscopic procedures easier [12]. Most conventional and simple overtubes, such as the Guardus overtube - gastric (US Endoscopy Group, Inc., Mentor, OH, USA), have been used to enable repeated endoscopic insertion. However, these simple overtubes are currently limited in use due to the high risk of intestinal perforation because of the materialŌĆÖs rigidity [12,14,15,17,19]. Most overtubes are short; hence, it is difficult to get into deep areas such as the small intestine.
We developed a guidetube that is made of silicone to overcome the disadvantages of the rigid types. Silicone is soft and flexible and can be safely used in many organs. Therefore, the new tube can be used more extensively and complications can be minimized. Insertion of the guidetube is safe and simple. There are 2 ways to insert it: First, the endoscope and guidetube are inserted together simultaneously. Second, the guidetube is inserted along with the endoscope.
The optimal length of the guidetube selected for the upper GI tract was 30 cm because insertion in the upper GI tract is relatively easy except in the hypopharynx, which is a mucosa-lined muscular tube surrounded by several muscle walls. Likewise, the optimal length of the guidetube selected for the lower GI tract was 30 cm because endoscopic insertion in the lower GI tract is relatively easy except for the junction of the sigmoid and descending colon. However, the length can vary based on the userŌĆÖs needs and patientŌĆÖs condition, such as a history of surgery or bowel deformity. In cases of multiple large polyps in the ascending colon, the optimal length of the guidetube is 70 cm. because the guidetube is inexpensive, users can adjust the guidetube length.
The new guidetube can be used to perform various procedures that were previously performed using an overtube, which presents many advantages. These include a reduction in the incidence of complications in the existing overtube, the ability for the operator to change the length as needed, and the low cost to manufacture, which means the guidetube can be used as a basis for all treatment endoscopes. In addition, complicated procedures can be performed by placing ancillary devices in the channels located within the guidetube. Table 2 shows endoscopic procedures performed easily and quickly through the guidetube.
This study had some limitations. First, all tests were performed in human GI simulators not in patients. Peristalsis, elasticity, and movement of a real human GI tract were not completely reproduced. Endoscopic procedures in a human GI simulator are easier to perform than in a real human GI tract. However, by choosing realistic simulators, this bias was minimized. Second, all tests were performed in easily accessible locations. However, the reason for choosing uncomplicated fixation positions was to exclude other factors affecting the endoscopic procedure time. Future studies in animals and humans should be conducted to overcome the limitations of the GI simulator in this study design.
In conclusion, all tests were performed quickly and safely using the new guidetube. Endoscopic procedures using the guidetube are simple and accessible methods for assisting the endoscopic removal of large multiple polyps and sharp foreign bodies in the GI tract. The guidetube can be used as a guide tunnel to approach the GI tract when a conventional approach is difficult. It is considered as an effective tool for improving the endoscopic approach with a low risk to patients compared with conventional approaches.