INTRODUCTION
Visceral artery pseudoaneurysms are rare and can be caused by atherosclerosis, pancreatitis, cholecystitis, inflammation, or trauma. These occur mainly in the splenic and hepatic arteries and rarely in the gastric artery, and they require urgent treatment as the mortality rates following their rupture are as high as 20ŌĆō40% [1]. Transcatheter arterial embolization is usually performed because it is safe and effective [2].
Complications after coil embolization include re-bleeding, infarction, abscesses, coil migration to other organs, and dissection of puncture vessel. Of these complications, coil migration to other organs after endovascular embolization is extremely rare, and only 16 cases have been reported previously [3-18].
We report a rare case of coil migration to the duodenal lumen after transcatheter arterial embolization of a right colic artery pseudoaneurysm. To the best of our knowledge, this is the first reported case of coil migration after embolization of a right colic artery pseudoaneurysm. Finally, we recommend a management approach for asymptomatic patients with coil migration.
CASE REPORT
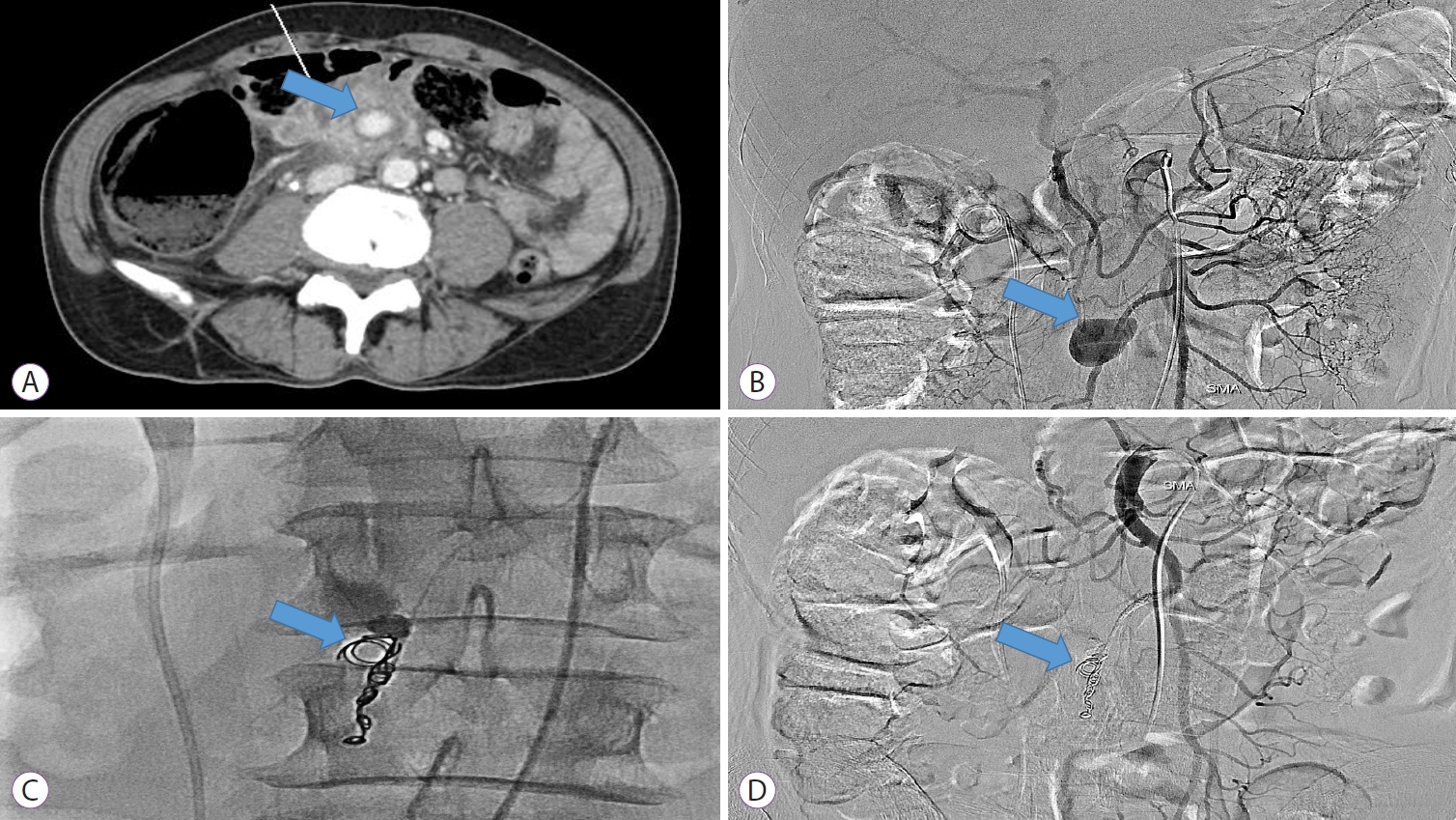
A 62-year-old man visited the emergency room with complaints of fever, vomiting, and epigastric pain since 1 week. The patient had hypertension and diabetes. A computed tomography (CT) scan revealed a pseudoaneurysm of the branch of the superior mesenteric artery and an organized hematoma in the distal small bowel (Fig. 1A). Angiography was then performed, showing a 4├Ś3 cm sized pseudoaneurysm of the right colic artery branch in the superior mesenteric artery (Fig. 1B). No contrast extravasation was observed. However, as the risk of rupture seemed high, we decided to perform embolization. Three coils were used for embolization therapy (Fig. 1C). Specifically, embolization coils (Tornado┬« embolization coil, 5 cm long, 0.018-inch diameter; COOK Medical LLC., Bloomington, IN, USA) were deployed at the distal end of the pseudoaneurysm. Next, embolization with a mixture of histoacryl and lipiodol was performed. After the procedure, superior mesenteric artery angiography showed no contrast filling in the pseudoaneurysm, confirming successful embolization (Fig. 1D). The patientŌĆÖs abdominal pain improved, and his vital signs stabilized.
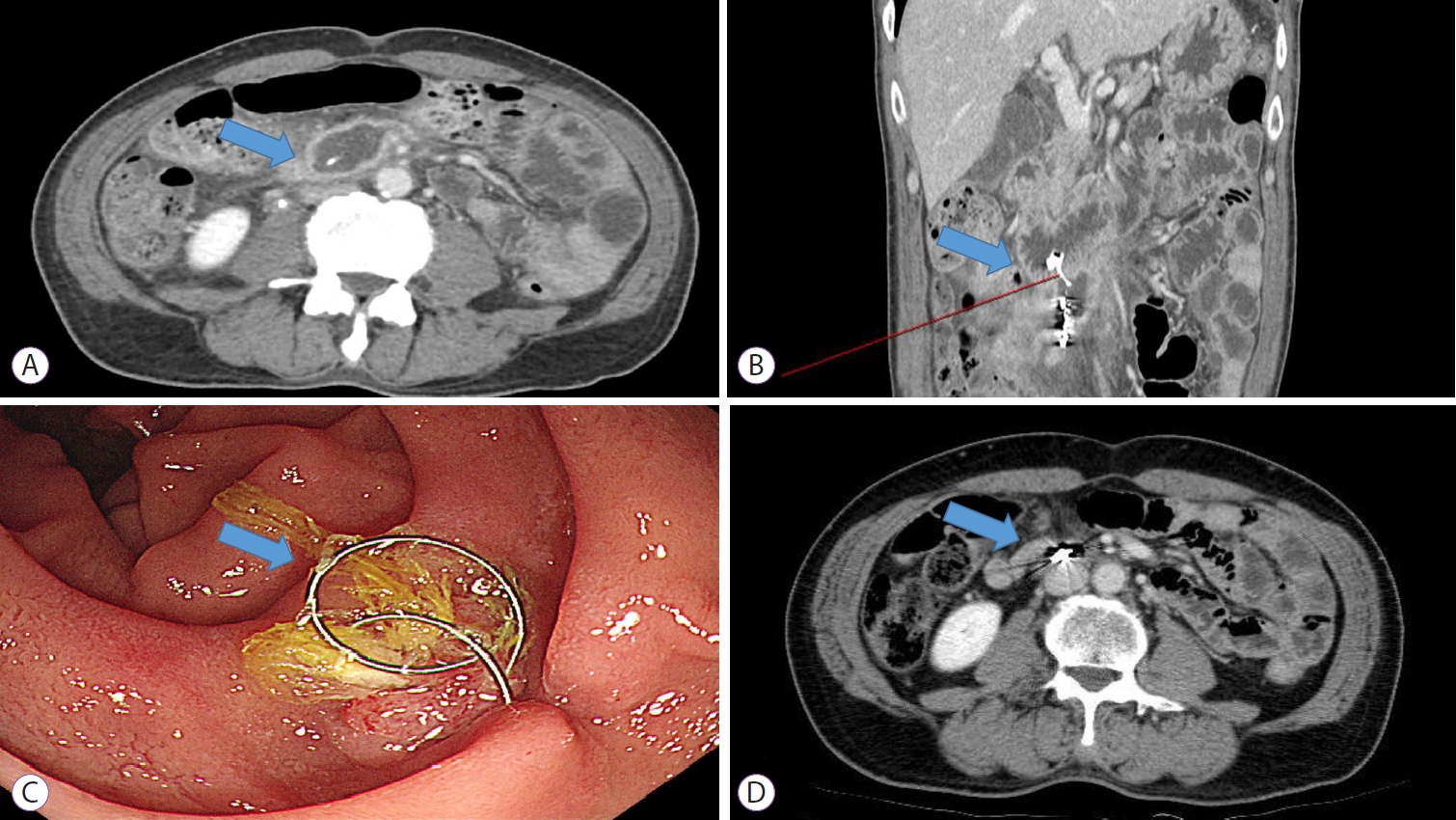
A follow-up abdominal CT six days after embolization showed penetration of coil material into the lumen of the third part of the duodenum (Fig. 2A, B). The patient had no fever, abdominal pain, or melena. He was carefully observed without further intervention and was discharged on the 16th day after embolization.
One month after discharge, an upper gastrointestinal endoscopy was performed to check if the coil had moved into the duodenal wall. It showed two strands of wire pulled out from the lumen inside of the third part of the duodenum that was covered with granulation tissue (Fig. 2C). The surrounding duodenal mucosa was normal, without necrosis or fistula. Initially, we tried to remove the coil using biopsy forceps; however, it was firmly attached to the duodenal wall. We believed it would be difficult and even harmful to remove the coil endoscopically since the coil was spiral-shaped. We decided to adopt a conservative approach by not removing the coil. Two months later, an abdominal CT was performed. The displaced coil was still seen in the duodenal wall (Fig. 2D). After 1 year of follow-up, no complications were observed.
DISCUSSION
We searched for previous case reports of coil migration after endovascular embolization for visceral artery pseudoaneurysm. Finally, a total of 17 cases, including our case, were reviewed [3-18]. Most patients had various underlying illnesses, including chronic pancreatitis or cholecystitis. The common sites of pseudoaneurysms were hepatic, splenic, and gastroduodenal arteries. The common migration sites were the stomach and common bile duct. Approximately 70% of patients showed obstructive signs and symptoms. Patients with coil migration to the common bile duct showed signs of cholangitis or had biliary colic pain.
Although the exact reason for coil migration is unclear, there are few reports about the risk factors associated with coil migration. Several reports mentioned the presence of infection [9], malignancy [3], or inflammation [18] around the pseudoaneurysm. Under these conditions, the coil may erode the thin walls of the pseudoaneurysm and migrate to another organ. As the sizes of the pseudoaneurysm varied dramatically, the number and sizes of the coils deployed also varied greatly across cases. The size of the coil used for embolization was found to be unrelated to the chance of its migration [4].
The duration between coil migration and embolization varied from several weeks to 10 years. In our patient, the migration developed unexpectedly early, i.e., 6 days after embolization. There is a possibility of intra-procedural coil migration. The very thin (0.05 mm) tip of the endovascular coil may damage the thin wall of the pseudoaneurysm and later penetrate into the adjacent organ. However, endovascular damage by the coil could not be identified in our study since angiography did not show extravasation after the embolization. In one study, an intra-procedural complication was reported, including rupture of the gastroduodenal artery by an endovascular coil [19].
Treatment for migrated coils has not been established yet, owing to the small number of cases. Among the 17 patients with coil migration, patients underwent endoscopic removal (n=4), surgical exploration (n=6) or combined endoscopic surgery (n=1), and conservative treatment (n=5). The remaining 1 patient had a fatal aortogastric fistula as a remote complication of embolization.
Patients whose coils migrated to the common bile duct underwent endoscopic retrograde cholangioscopic removal of the migrated coils [7,11], percutaneous cholangioscopic extraction [15], or open surgical exploration [16,17]. One patient with coil migration to the jejunum underwent upper endoscopic removal [8]. Hot biopsy forceps were used for the removal of the thick coil wires [8]. Most patients had good treatment outcomes.
Treating patients with asymptomatic coil migration is challenging because little is known about the long-term course of natural disease. In 3 out of 5 asymptomatic patients, the migrated coils passed spontaneously through the stomach [3], rectum [13], or ureterovesical junction [12]. Matsubara et al. [3] reported on coil migration into the stomach after embolization for splenic artery pseudoaneurysm. The patient was carefully followed-up, because there was no active bleeding or peritonitis, and the coil was excreted spontaneously 6 months later. Our patient had no signs of obstruction, fistula, or any other symptoms. The migrated coil penetrated the duodenal wall that was covered with granulation tissue and a normal surrounding mucosal layer. As it was hard to remove it endoscopically, the patient was followed up and no complications were observed for 12 months.
In conclusion, we reported a rare case of coil migration to the duodenal lumen after embolization of a right colic artery pseudoaneurysm. The patient showed no symptoms and was treated conservatively without any intervention. Our case supports conservative treatment as the primary treatment for asymptomatic patients. Clinicians should assess the risks and benefits of coil removal in asymptomatic patients, prior to performing an intervention.