Increased Detection of Colorectal Polyps in Screening Colonoscopy Using High Definition i-SCAN Compared with Standard White Light
Article information
Abstract
Background/Aims:
The aim of this study was to evaluate the efficacy of high definition (HD) i-SCAN for colorectal polyp detection in screening colonoscopy.
Methods:
We retrospectively analyzed the records of 501 patients who had undergone screening colonoscopy performed by three endoscopists with either HD i-SCAN (n=149) or standard white light (n=352) from January 2, 2014 through June 30, 2014. Patient information and inter-endoscopist variation as well as polyp number, endoscopic findings, and pathologic characteristics were reviewed.
Results:
The detection rates of colorectal and neoplastic polyps were significantly higher using HD i-SCAN than standard white light colonoscopy (52% vs. 38.1%, p=0.004 for colorectal polyps; and 37.2% vs. 27.9%, p=0.041 for neoplastic polyps). Analysis of endoscopic findings revealed no difference in detected polyp size between HD i-SCAN and standard white light colonoscopy (4.59±2.35 mm vs. 4.82±2.81 mm, p=0.739), but non-protruding polyps were more commonly detected by i-SCAN than by standard white light colonoscopy (24.6% vs. 13.5%, p=0.007).
Conclusions:
Colonoscopy using HD i-SCAN had a significantly higher detection rate of colorectal polyps, including neoplastic polyps, because of improved sensitivity for detecting non-protruding lesions.
INTRODUCTION
Colonoscopy is considered the optimal examination method for detecting colorectal polyps, particularly in view of the ability to provide therapeutic polypectomy in conjunction with diagnosis. However, colonoscopy is not perfect. The rate of interval cancer is 3% to 6% [1,2], which has been inversely linked to adenoma detection rates using screening colonoscopy [3]. Indeed, colonoscopic miss rates range from 6% to 12% for adenomas larger than 1 cm [4,5].
As concerns increase about the effectiveness of colonoscopy for detection of adenomas and early-stage colorectal cancer, new endoscopic techniques have been introduced. Chromoendoscopy and cap-assisted colonoscopy show benefits over standard colonoscopy for colorectal polyp detection [6,7]. However, these methods are not suitable for all colonoscopy cases. Chromoendoscopy is inconvenient due to the need for dye spraying and the suction of collected dye in the colon, and cap-assisted colonoscopy requires extra equipment to be added onto standard colonoscopes. Other technologies such as capsule colonoscopy, magnification endoscopy, and confocal laser endomicroscopy have been used to increase detection of colorectal polyps, but their value in screening has yet to be determined [8,9].
Virtual chromoendoscopy, including narrow-band imaging (NBI; Olympus, Tokyo, Japan), Fujinon imaging color enhancement (FICE; Fujinon, Tokyo, Japan), and i-SCAN (Pentax, Tokyo, Japan) are used for detecting and characterizing subtle epithelial lesions in the gastrointestinal tract [10]. These virtual chromoendoscopes are equipped with a push-button system that enables rapid switching between white light (WL) images and computer-enhanced images. They have shown promising efficacy for predicting the real-time histology of epithelial lesions [10,11]. However, most of the studies using NBI or FICE did not show a significant improvement in detection of colorectal polyps [12-15].
The efficacy of colorectal polyp detection using i-SCAN has not been established since there have been very few studies [16-20]. The aim of this study was to determine the usefulness of i-SCAN for the detection of colorectal polyps via a comparison of colonoscopy using high definition (HD) i-SCAN to standard WL colonoscopy.
MATERIALS AND METHODS
Study population and data collection
The present study was performed on 1,957 persons who received colonoscopy during medical checkups at the Health Promotion Center of Hallym Hospital between January 2, 2014 and June 30, 2014. A total of 501 cases were defined as screening colonoscopy using the following inclusion criteria: (1) age 40 to 75 years; (2) no history of gastrointestinal signs, symptoms, and/or relevant diagnoses; (3) no history of colon cancer, polyps, and/or gastrointestinal disease; (4) no family history of colorectal cancer; and (5) no history of colorectal examination, including sigmoidoscopy, colonoscopy, and barium enema, within the previous 5 years. All colonoscopies were performed by three experienced endoscopists certified by the Korean Society of Gastrointestinal Endoscopy.
The data were analyzed retrospectively using electronic medical records, colonoscopy reports and images, and pathology reports. Colonoscopy reports included the name of the endoscopists; the type of instruments; whether or not it was a complete colonoscopy; intubation and withdrawal time; bowel preparation quality; endoscopic characteristics of detected colorectal polyps, including number, size, location, and morphology according to the Paris classification [21]; and polyp removal method. In reviewing the characteristics of detected colorectal polyps, we simultaneously investigated endoscopic images and colonoscopy and pathology reports to identify whether polyps were detected during the intubation period. Polyps or cancers detected during the intubation period were excluded from the analysis. The polyp detection rate was defined as the proportion of patients with at least one polyp.
In additional reviews of endoscopic and pathologic findings, all detected polyps were analyzed. Endoscopic morphologies of colorectal polyps were separated into protruding lesions, including sessile, semi-pedunculated and pedunculated, and non-protruding lesions. Neoplastic polyps were defined as low-grade dysplasia, high-grade dysplasia, tubulovillous, serrated adenoma, and carcinoma in situ.
The study was approved by the Institutional Review Board at Hallym Hospital.
Colonoscopy equipment
HD i-SCAN colonoscopies were performed with EPK-i5000 (Pentax EPKi processor) and EC38-i10F model colonoscopes (Pentax). The i-SCAN has three distinct modes of image enhancement. Surface enhancement (SE) highlights structure through recognition of the edges, and contrast enhancement (CE) highlights depressions and structural differences through colored presentation of low-density areas. Tone enhancement (TE), similar to NBI, dissects and analyzes the individual red, green, and blue components of an ordinary endoscopic image and enhances the image by recombining altered color frequencies [22]. The i-SCAN can be used in a single mode or combined modes. SE and CE each have three enhancement levels (2+, 3+, and 4+), and TE has six modes for pit pattern, vessel, Barrett’s, esophagus, stomach, and colorectum. Since increased enhancement using SE and CE modes results in heightened endoscopic imaging noise, settings needed to be optimized for general application. After a 2-month introductory period in our hospital, we decided that the optimal i-SCAN mode settings for colorectal polyp detection were 2+ for SE, 2+ for CE, and colorectum mode for TE. When HD i-SCAN was used, the colonoscope was inserted into the cecum base in the WL mode. The withdrawal phase was then performed using the i-SCAN mode.
Standard WL colonoscopies were performed with an EVIS LUCERA CV-260 processor, EVIS LUCERA CLV-260 light source, and CF type H260AL/I colonoscopes (Olympus).
Colonoscopy procedure
Three endoscopy units are available for patients undergoing screening colonoscopy in the Health Promotion Center. Two are equipped with standard WL colonoscopy settings and one with HD i-SCAN colonoscopy settings. Three endoscopists regularly rotated through the two endoscopic techniques depending on availability of the instruments.
All patients received standard bowel preparation, which included 2 L of polyethylene glycol (Coolprep powder; Taejun, Seoul, Korea) and 2 L of free water at home. Bowel preparation quality was assessed using the Aronchick bowel preparation scale [23]. The three endoscopists followed the rules of the Hallym endoscopy unit as follows: (1) the minimal withdrawal time was at least 6 minutes; (2) endoscopists intubated the cecum quickly and confirmed cecal intubation by documenting a cecal landmark such as the cecal valve or appendix orifice; (3) the size of all polyps was measured by open biopsy forceps; and (4) all detected polyps were directly removed during the procedure using biopsy or polypectomy.
Statistical analysis
All analyses were performed using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). The chi-square test or Fisher exact test were performed for categorical variables. Quantitative data were summarized using mean values and standard deviations (SD), and the Mann-Whitney test was performed to detect significant differences. A p<0.05 indicated a significant difference.
RESULTS
Baseline characteristics and colonoscopy quality assessment
Base line characteristics of the study population and colonoscopy quality indicators are summarized in Table 1. A total of 501 colonoscopies were evaluated (HD i-SCAN, n=149; standard WL, n=352). Nine cases were excluded due to poor bowel preparation (HD i-SCAN, n=1; standard WL, n=8). The study population was predominantly male (56.3%) and the mean±SD was 51.2±11.73 years. There was no significant difference in sex, age, or bowel preparation between the two groups. The withdrawal time (mean±SD, range) was longer using HD i-SCAN (7.18±2.54 minutes [range, 6 to 30]) than standard WL colonoscopy (6.80±1.75 minutes [range, 6 to 25]; p=0.007). However, in further analysis of the withdrawal time according to whether colon polyps were detected or not, there were no significant differences between the two groups.
Three endoscopists performed 189, 141, and 162 colonoscopies, respectively. There was no significant difference between the proportion of times each used HD i-SCAN (31.7%, 30.5%, and 27.8% of the time, respectively, p=0.716). Polyp detection rates were 43.9% vs. 38.3% vs. 43.3% (p=0.527) for colorectal polyps, and 30.7% vs. 28.4% vs. 32.7% (p=0.715) for neoplastic polyps, respectively. There were no significant differences between the three endoscopists (Table 2).
Colorectal polyp and neoplastic polyp detection rate
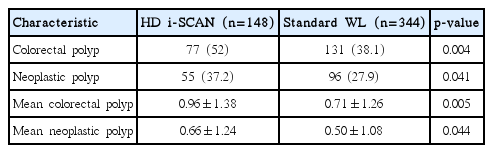
HD i-SCAN colonoscopy showed that 77 of the 148 subjects had colorectal polyps and that 55 of the 77 patients with polyps had at least one neoplastic polyp. Polyp detection rates were significantly higher using HD i-SCAN than standard WL colonoscopy (52% vs. 38.1%, p=0.004 for overall colorectal polyps detected; 37.2% vs. 27.9%, p=0.041 for neoplastic polyps detected) (Table 3). A total of 363 polyps were identified during the withdrawal phase. HD i-SCAN and standard WL colonoscopy detected 134 and 229 polyps, respectively. A significantly higher mean number of colorectal and neoplastic polyps were detected by HD i-SCAN than standard WL colonoscopy (0.96±1.38 vs. 0.71±1.26, p=0.005 for colorectal polyps; 0.66±1.24 vs. 0.50±1.08, p=0.044 for neoplastic polyps).
Characteristics of endoscopic and pathologic findings of identified colorectal polyps
Table 4 shows the detailed endoscopic and pathologic characteristics of the detected polyps. The mean polyp sizes (±SD) using HD i-SCAN and standard WL colonoscopy did not show significant differences (4.59±2.35 mm vs. 4.82±2.81 mm, p=0.739). However, non-protruding polyps were more commonly detected by HD i-SCAN than by standard WL colonoscopy (24.6% vs. 13.5%, p=0.007). There was no significant difference in colorectal polyp location between the two groups. Pathologic findings showed that the proportion of neoplastic polyps detected was not different between HD i-SCAN and standard WL colonoscopy (69.4% vs. 69.9%, p=0.926).
DISCUSSION
Virtual chromoendoscopies have been proposed to lead to better recognition of small or flat lesions, but the absolute increase in diagnostic yield seems limited. NBI, one of the most widely used and extensively studied image enhancement technologies, did not show a clear benefit for the detection of colorectal polyps compared with standard or HD WL colonoscopy [12,13,24]. In contrast, NBI is useful for detecting lesions in small lumens, such as the esophagus [25,26]. This difference might be caused by the fact that NBI images are much darker than WL images, particularly in regions like the colon and rectum that have a large luminal diameter. In contrast, i-SCAN is as bright as standard WL and could therefore be utilized for observing much larger areas than NBI and for improved detection of colorectal polyps [22]. However, the effectiveness of i-SCAN for colorectal polyp detection is not yet clear because of insufficient data.
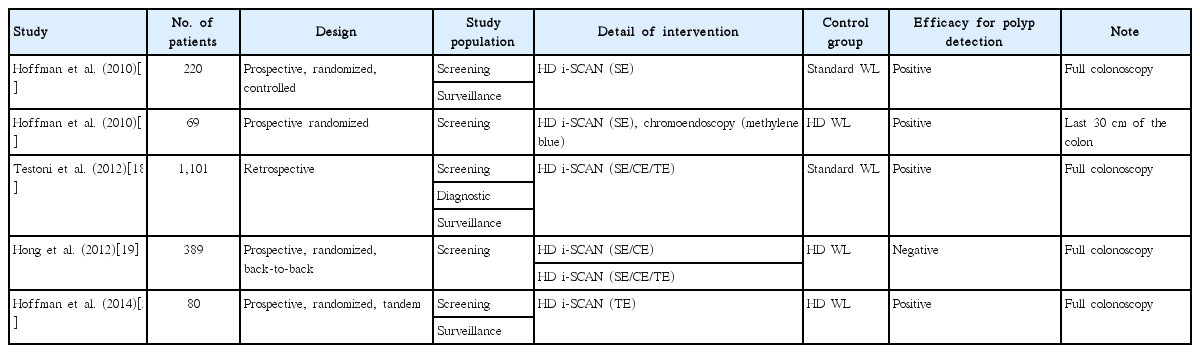
To date, only five studies have been published evaluating the efficacy of i-SCAN for colorectal polyp detection (Table 5). The heterogeneous methods used in these studies made it difficult to confirm the efficacy of i-SCAN for colorectal polyp detection. For this reason, additional data collection and studies are required.
The present study, even though retrospective, utilized a relatively large number of patients who underwent screening colonoscopies. In the present study, HD i-SCAN was significantly better at detecting colorectal and neoplastic polyps compared with standard WL colonoscopy. However, based on pathologic characteristics, the proportion of neoplastic polyps detected using HD i-SCAN vs. standard WL was nearly the same. Analysis of endoscopic characteristics of the detected polyps revealed that HD i-SCAN had a higher sensitivity of detecting non-protruding polyps. Therefore, it was suspected that the improved neoplastic polyp detection rate of HD i-SCAN resulted from its increased ability to detect non-protruding polyps, leading to an overall higher detection rate for all colorectal polyps, including neoplastic, hyperplastic, and inflammatory polyps. Colorectal polyp morphology, especially flat lesions, is a major factor influencing missed detection of polyps [27]. In agreement with the present study, previous studies revealed the effectiveness of HD i-SCAN for detecting non-protruding lesions.
Other factors that influence colorectal polyps remaining undetected are poor colon cleansing [28], experience level of the endoscopist, and longer withdrawal time [29]. In the present study, we took into account the differences in bowel preparation quality, withdrawal time, and interendoscopist variation including colorectal and neoplastic polyp detection rate, and usage rate between HD i-SCAN and standard WL colonoscopy. Although the withdrawal time for the HD i-SCAN was longer than for standard WL colonoscopy, this included the time it took to complete procedures, such as biopsies and polypectomies. Since more colorectal polyps were detected using HD i-SCAN, more time was required to remove polyps than with standard WL colonoscopy. If we had spent more time detecting colon polyps with HD i-SCAN than with standard WL colonoscopy, the withdrawal time might be longer, even when we did not detect polyps. However, when patients were grouped according to whether colon polyps were detected, further analysis of the withdrawal time (regardless of polyp detection) did not show a difference between HD i-SCAN and standard WL colonoscopy. These results indicated that the difference in withdrawal time resulted from procedure time relative to polyp detection, rather than an allowance of extra time specifically for HD i-SCAN colonoscopy.
Adenoma detection rate is an important measure of high-quality endoscopy. An older version of the guideline suggests adenoma detection rates of at least 25% for men and 15% for women at average risk [30], and a more recent study reported adenoma detection rates of 33.7% [31]. A simple comparison should not be made between adenoma detection rates of previous studies and the neoplastic polyp detection rate of the present study; with respect to neoplastic polyps, including adenomas and carcinoma in situ, which were detected only during withdrawal, the neoplastic polyp detection rates of HD i-SCAN and standard WL colonoscopy were similar or higher than in a previous study.
New imaging technology, including i-SCAN, can permit the endoscopist to be more focused during procedures than usual, and will probably promote greater accuracy in polyp detection; however, there is a significant learning curve for its use, even by an expert. To eliminate this bias, we used a 2-month introductory period to identify an optimal i-SCAN mode setting, and the three endoscopists respectively performed 111, 95, and 87 procedures with HD i-SCAN during the introductory period, including screening, surveillance, and diagnostic colonoscopy.
It is possible that HD imaging quality influenced the polyp detection rate independently of i-SCAN usage. Although HD colonoscopy provides better imaging, it is not clear that HD colonoscopy is a better diagnostic tool for colorectal polyp detection. There is some evidence that HD colonoscopy combined with virtual chromoendoscopy, especially NBI, improved detection of colorectal polyps compared with standard WL colonoscopy alone [32,33], but the majority of the studies evaluating polyp detection using HD compared to standard WL colonoscopies did not show increased efficacy [34,35]. In the present study, we could not exclude the possibility that HD added value to the i-SCAN compared to standard WL colonoscopy. A prospective, large-scale study is needed to further characterize the usefulness of HD i-SCAN chromoendoscopy.
In conclusion, our study showed that HD i-SCAN, a newly developed, virtual chromoendoscopy technique, improved detection of colorectal polyps in screening colonoscopy via increased sensitivity for non-protruding polyps. We suggest that increased use of HD i-SCAN chromoendoscopy in clinical practice could lead to more neoplastic polyp detection.
Notes
Conflicts of Interest:The authors have no financial conflicts of interest.