AbstractBackground/Aims:This study aimed to evaluate the prevalence and natural progression of subepithelial lesions (SELs) in the upper gastrointestinal (UGI) tract.
Methods:The medical records of patients with UGI SELs who underwent endoscopic screening at eight university hospitals between January and December 2010 were retrospectively investigated. The follow-up evaluations were performed until December 2016.
Results:UGI SELs were found in 1,044 of the 65,233 participants screened (endoscopic prevalence, 1.60%; the total number of lesions, 1,062; mean age, 55.1┬▒11.2 years; men, 53.6%). The median follow-up period was 48 (range, 8ŌĆō74) months. SELs were most frequently found in the stomach (63.8%) and had a mean size of 9.9┬▒6.1 mm. Endoscopic ultrasonography (EUS) was performed in 293 patients (28.1%). The most common lesions were leiomyomas, followed by gastrointestinal stromal tumors (GISTs), and ectopic pancreas. The proportions of SELs with malignant potential according to size were 3% (<1 cm), 22% (1ŌĆō2 cm), 27% (2ŌĆō3 cm), and 38% (Ōēź3 cm). In gastric SELs larger than 1 cm, resections were performed in 20 patients because of an increase in size, of which 12 were found to be GISTs.
INTRODUCTIONSubepithelial lesions (SELs) in the gastrointestinal (GI) tract include lesions with a smooth prominence in the inner cavity of the GI tract and no changes in the mucosal surface on endoscopy. SELs are subepithelial tumors in the GI wall layer that grow under the epithelium. SELs are typically classified as non-neoplastic or neoplastic. Most lesions are asymptomatic and clinically insignificant. However, neuroendocrine tumors (NETs), lymphomas, glomus tumors, and gastrointestinal stromal tumors (GISTs) are often malignant or potentially malignant. Despite state-of-the-art endoscopic facilities and extensive research, SELs are still difficult to diagnose before surgery using noninvasive methods, such as endoscopy or endoscopic ultrasound (EUS). Histological diagnosis of lesions includes bite-on-bite biopsy, EUS-guided fine needle aspiration/biopsy (FNA/B), endoscopic mucosal resection/endoscopic submucosal dissection, and submucosal tunneling with endoscopic resection.1-3 To detect and treat malignant lesions early, appropriate diagnostic approaches are required according to the characteristics of the lesions.4,5
Globally, there are few reports on the prevalence of upper GI SELs. Endoscopic identification has increased in recent years owing to the development of endoscopic technologies and increased interest in upper GI SELs.6 In Korea, the incidence of upper GI SELs from a single-center study was reported to be 0.8% to 1.7%, but these data are insufficient; there are no established guidelines, and hence each hospital uses a variety of approaches.7,8
The current clinical guidelines suggest that hypoechoic lesions in the second or third layers are removed if they are symptomatic or larger than 2 cm; even in the absence of symptoms, as much of the pathology is obtained as possible.9 On the other hand, SELs on the fourth layer are recommended for removal unconditionally due to the possibility of GISTs.4 EUS images often cannot distinguish malignant lesions and third-layer tumors. In Korea, esophagogastroduodenoscopy (EGD) is frequently performed, and endoscopic resections have also been developed.10-12 Therefore, it is necessary to assess whether the guidelines are followed in actual clinical conditions. We investigated the incidence, clinical practice patterns (diagnostic approaches), and the natural progression of upper GI SELs.
METHODSSubjectsThis multicenter retrospective study was conducted between 2010 and 2016 at the EUS Research Group of the Korean Gastrointestinal Endoscopy Society. The medical records of patients with upper GI SELs among the population who underwent endoscopic screening at eight university hospitals between January 1 and December 31, 2010, were retrospectively analyzed. These participants, in whom SELs were found, were followed up for 6 years through to 2016. Gastroscopy and EUS reports and clinical courses were reviewed.
DefinitionsDiagnoses were divided into clinical and pathological. Pathological diagnoses were made using endoscopic biopsy, EUS-guided FNA/B, endoscopic resection, or surgical resection. If a pathological diagnosis could not be obtained, a clinical diagnosis was made by tracking the clinical progress for more than 1 year and ultrasound imaging.
Examinations and endpointsFirst, for each healthcare center, the number of patients with SELs found during gastroscopy was checked in those who had undergone endoscopy for 1 year, and the number of patients who received additional tests, such as EUS, computed tomography, and biopsy. In addition, the percentage of patients who underwent endoscopic or surgical resection was determined, and those who did not undergo resection were followed up until 2016. During follow-up, the number and interval of follow-ups, size change of the SEL, and presence or absence of tumor resection were identified.
The primary endpoint was the incidence of upper GI SELs in asymptomatic patients. The secondary endpoints were clinical practice patterns and natural courses.
RESULTSPrevalence and characteristics of upper GI SELsBetween January 1 and December 31, 2010, 65,233 individuals underwent screening endoscopies at eight university hospitals. Upper GI SELs were identified in 1,044 patients (1,062 lesions in total). Thus, the endoscopic prevalence of upper GI SELs was thus 1.60%. The prevalence differed significantly between institutions (range, 0.72%ŌĆÆ3.48%; Table 1). The median age of the participants was 56 years (range, 12ŌĆÆ86 years, mean 55.1┬▒11.2 years), and 560 participants (53.6%) were men. Eighteen patients (1.7%) had two or more tumors. If an external indentation or vascular structure could be distinguished by endoscopy or other imaging studies, it was excluded from the SELs.
The most common location was the upper third of the stomach (39.9%). Ten of the 14 histologically confirmed GISTs were located in the upper body. The mean size of the SELs when they were first identified on endoscopy was 9.9┬▒6.1 mm, and 49.1% of the SELs were 1 cm or larger, 7.0% were larger than 2 cm, and 1.7% were larger than 3 cm (Table 2).
Among the 1,044 patients with SELs identified by EGD, 293 (28.1%) underwent additional endoscopic ultrasonography. The EUS characteristics of the upper GI SELs according to the organ are described in Table 3. A total of 154 lesions (14.8%) were biopsied. The histological results of the biopsy confirmed were as follows; esophageal duplication cyst (1), esophageal or gastric leiomyoma (11), esophageal hemangioma (1), gastric or duodenal ectopic pancreas (10), gastritis cystica profunda (1), inflammatory fibrinoid polyp (1), lipoma (1), gastric adenocarcinoma (1), BrunnerŌĆÖs gland hyperplasia (7), duodenal lymphangioma (2), and duodenal neuroendocrine tumor (1).
A total of 394 SELs were followed up for >12 months, with a median of 54 months (range, 12ŌĆÆ83 months); of these, the esophagus, stomach, and duodenum SELs were followed up 59 (range, 12ŌĆō77) months, 52 (range, 12ŌĆÆ83) months, and 60 (range, 12ŌĆÆ82) months, respectively. Fifty-four patients (5.2%) had their SELs removed by endoscopic procedures or surgery, 29 immediately after diagnosis, and 25 during follow-up, owing to increased SEL sizes (Fig. 1). The final histological results of the 54 resected SELs were as follows: GIST (11); leiomyoma (10); ectopic pancreas (10); NET (3); granular cell tumor (3); Brunner's gland adenoma (3); lipoma (2); inflammatory fibroid polyp (2); schwannoma (1); advanced gastric cancer (1); lymphoepithelioma-like carcinoma (medullary carcinoma) (1); eosinophilic abscess (1); ectopic gastric mucosa (1); and not mentioned (5)
Types and distribution of SELs according to the locationThe most common SELs among all the upper GI SELs were leiomyomas (30.4%), followed by GISTs (24.6%), ectopic pancreas (16.7%), and lipomas (5.1%) (Table 4). The most common SELs in the stomach were GIST (39.1%), ectopic pancreas (22.5%), and leiomyoma (18.3%). Common SELs in the esophagus were leiomyoma (75.8%) and granular cell tumor (15.2%), whereas common SELs in the duodenum were Brunner's gland hyperplasia/adenoma (29.3%) and lymphangioma (22.0%).
Some SELs were uniquely distributed in the esophagus and duodenum. For example, granular cell tumors (15% of esophageal SELs) occur in the esophagus. Brunner's gland hyperplasia/adenoma (29% of duodenal SELs) and lymphangiomas (22% of duodenal SELs) were observed in the duodenum.
Forty-four esophageal and gastric SELs were greater than 2 cm in size. Among them, the proportion of malignant SELs was 18% (seven GISTs and one adenocarcinoma; Fig. 2). In the stomach, the proportion of malignant SELs increased with size, with 3% of SELs >1 cm, 29% of SELs >2 cm, and 38% of SELs >3 cm.
Natural course of upper GI SELs
Figure 3 shows the clinical practice patterns of gastric SELs greater than 1 cm and gastric SELs greater than 2 cm. Further, 34 stomach SELs measured >2 cm in size. Eight SELs were pathologically diagnosed, and 3 SELs proved to be malignant (one adenocarcinoma and two GISTs). Additionally, 22 SELs were followed up without pathological confirmation (median, 48 months; range, 8ŌĆō74 months). Seven gastric SELs >2 cm increased in size during a median follow-up period of 60 months (range, 45 to 74 months) and were resected endoscopically or surgically. Fifteen SELs remained unchanged in size or shape during the median follow-up period of 46 months (range, 8ŌĆō61 months). Eventually, 65% of gastric SELs >2 cm were clinically followed up without a pathological diagnosis; among them, 13.6% (3/22) were found to be malignant.
Figure 4A shows the practice patterns of the 246 esophageal SELs. The pathological analysis confirmed leiomyomas in 17 patients. A total of 105 (42.7%) patients were followed up without pathological confirmation. Four patients underwent SEL resection because of an increase in size.
Approximately 37 of the 132 patients with duodenal SELs attempted to obtain a pathological diagnosis (Fig. 4B). Twenty patients were pathologically confirmed at their first detection, and two were diagnosed with endoscopic resection of an NET. Among those with confirmed duodenal SEL, 33 (25.0%) were followed up without a pathological diagnosis. Four SELs that had increased in size during the follow-up period were resected. The malignant potential was observed in 1.5% (2/132) of duodenal SELs (duodenal NETs).
A total of 35 SELs (seven esophageal, 24 gastric, and four duodenum SELs) increased in size during the median 58 months follow-up period (range, 12ŌĆō73 months; mean, 51.5┬▒17.5 months). The size of SELs was 1.3┬▒0.4 cm at the initial diagnosis and 2.0┬▒0.8 cm at the last follow-up or excision. The pathologies were GIST in five patients, leiomyoma in three patients, ectopic pancreas in one patient, and others that could not be identified.
DISCUSSIONThis study evaluated the prevalence and natural progression of upper GI SELs detected during endoscopic screening. We also evaluated the clinical practice patterns of upper GI SELs. The prevalence in the total screening population was 1.6% (1,044 out of 65,233). The institutional prevalence varied from 0.72% to 3.48%. In previous studies, the prevalence of upper GI SELs in the screening population was 0.8% to 1.7%.7,8 Studies performed in the Korean population were 0.8% to 1.1% during 2010ŌĆō2022. These large differences between institutions suggest that the detection of upper GI SELs may depend more on the fidelity of the endoscopist or endoscopic records than on an increase in the actual prevalence or development of endoscopic techniques.
In this study, 54 SELs were endoscopically or surgically resected immediately after diagnosis, and 28 SELs were resected during follow-up owing to their increased size. Considering that the SELs lost during follow-up were not counted, there is likely to be an increase in tumor size. Several studies investigated the natural history of upper GI SELs. Lim et al.7 reported an increase in the size of SELs in only 8 (3.2%) of 252 SELs over 84 months.Other studies showed that only 8.5% of 989 SELs changed significantly in size and morphology at a median follow-up period of 24 months (range, 3ŌĆō123 months).
Most SELs are benign at diagnosis. NETs, lymphomas, glomus tumors, and GISTs are generally malignant or likely to become malignant. Endoscopy makes it difficult to predict malignant tumors based solely on the tumor shape. A possible diagnosis can be made by synthesizing endoscopic characteristics, such as tumor location, organ, mobility, and consistency.13 In general, additional diagnostic modalities of EUS are required. In this study, only 28.1% (293/1,044) of the SELs underwent additional EUS. This may have included some SELs already discovered before the study and those undergoing follow-up. The common frequency of tumors by organ was not significantly different from that reported in previous studies. GISTs are the most common SELs of the stomach.
Small, incidentally detected SELs can be monitored periodically using endoscopy or EUS. The European Society of Gastrointestinal Endoscopy guidelines recommend that the management of each SEL depends on its size, location, pathology, and malignant potential or symptoms.14,15 A pathological diagnosis is recommended if the lesion is larger than 2 cm or if EUS suggests malignancy.16 Several SELs, such as lipomas, cysts, or ectopic pancreas, show typical characteristics during endoscopy or EUS. However, the diagnostic accuracy of EUS for SELs located in the third and fourth layer is unsatisfactory without pathology. Tissue acquisition for histopathological diagnosis is often necessary to decide further treatment policies.17-19 SELs arising from the submucosa and muscularis propria can be sampled using tunnel biopsies (or deep-well biopsies), EUS-guided FNA/B, or advanced endoscopic techniques (unroofing or endoscopic resection).2,20-25
This study has some limitations. First, the prevalence rates varied among different institutions, suggesting that the incidence may depend on the endoscopist. Second, because this was a retrospective study, it was impossible to review all endoscopic images. Pathological diagnosis, which is the gold standard, was not performed for all lesions; therefore, it was impossible to calculate an accurate diagnosis.
In this study, the tissue yield for the pathological diagnosis of upper GI SEL was not high at approximately 44%. Therefore, it is important to follow up according to clinical judgment using endoscopy and EUS findings, as well as a biopsy. In this study, in the stomach, the proportion of malignant SELs increased as the size increased, and in the case of upper GI SELs of size 1 cm or larger, it is necessary to recommend additional tests, such as biopsy or EUS. If the pathology cannot be confirmed, close follow-up may be needed.
In conclusion, the prevalence of SELs in the upper GI tract was 1.60% in an endoscopically screened population. In the case of the stomach, where GIST occurs, it is necessary to recommend additional tests such as biopsy or EUS if the size is Ōēź1 cm
NOTESFig.┬Ā1.Study population. There were 29 cases of subepithelial lesions resected immediately after diagnosis, and 25 cases resected after follow-up gastroscopy. 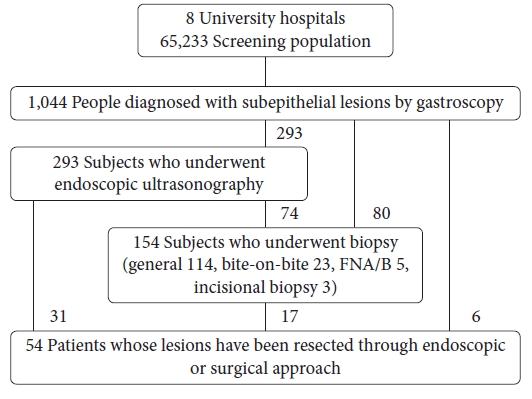
Fig.┬Ā2.Distribution of upper gastrointestinal subepithelial lesions (SELs) according to size. (A) Types of SELs of 2 cm or more. (B, C) Comparison of the proportion of malignancy or premalignant gastric SELs by size. Malignancy or premalignant lesions included gastrointestinal stromal tumors, neuroendocrine tumors, and gastric cancer. BGH, BrunnerŌĆÖs gland hyperplasia; Ext., external; GIST, gastrointestinal stromal tumor. 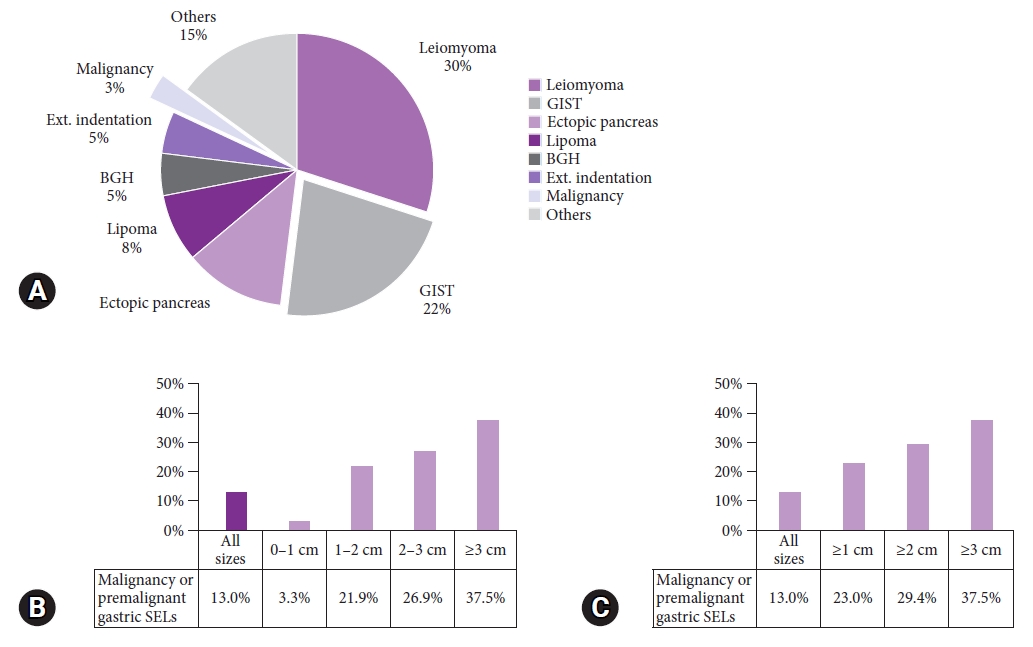
Fig.┬Ā3.Natural course of gastric subepithelial lesions (SELs) according to size. (A) Gastric SELs measuring 1 to 2 cm in size. (B) Gastric SELs larger than >2 cm. Bx., biopsy; GCP, gastritis cystica profunda; IFP, inflammatory fibroid polyp; FNA/B, fine needle aspira┬Łtion/biopsy; ER, endoscopic resection; Eo., eosinophilic; GIST, gastrointestinal stromal tumor; AGC, advanced gastric cancer. 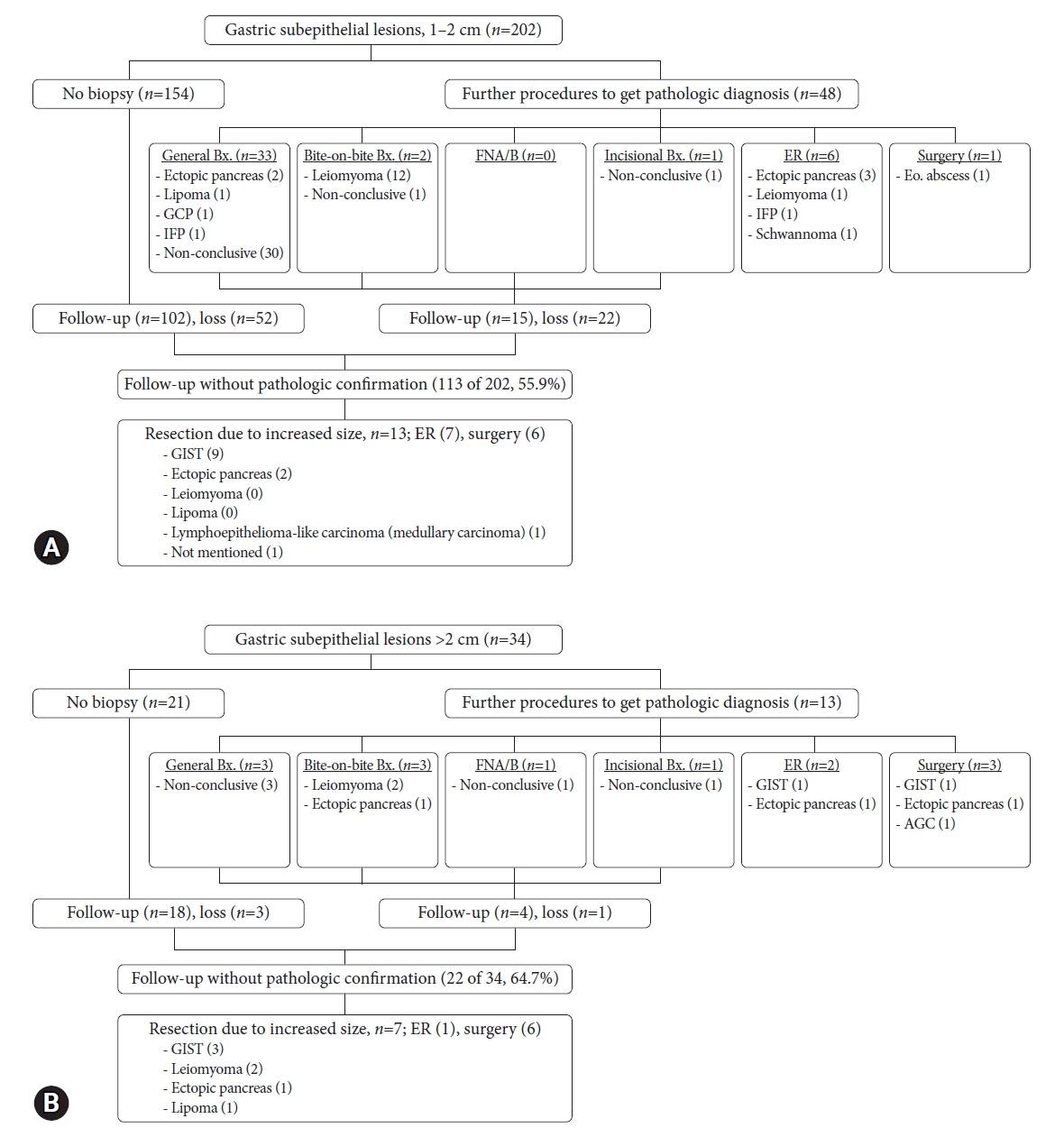
Fig.┬Ā4.Clinical practice and natural course of (A) esophageal and (B) duodenal subepithelial lesions. Bx., biopsy; ER, endoscopic resection; GCT, granular cell tumor; GIST, gastrointestinal stromal tumor; F/U, follow-up; BGA, BrunnerŌĆÖs gland adenoma; BGH, BrunnerŌĆÖs gland hyperplasia; NET, neuroendocrine tumor; FNA, fine needle aspiration. 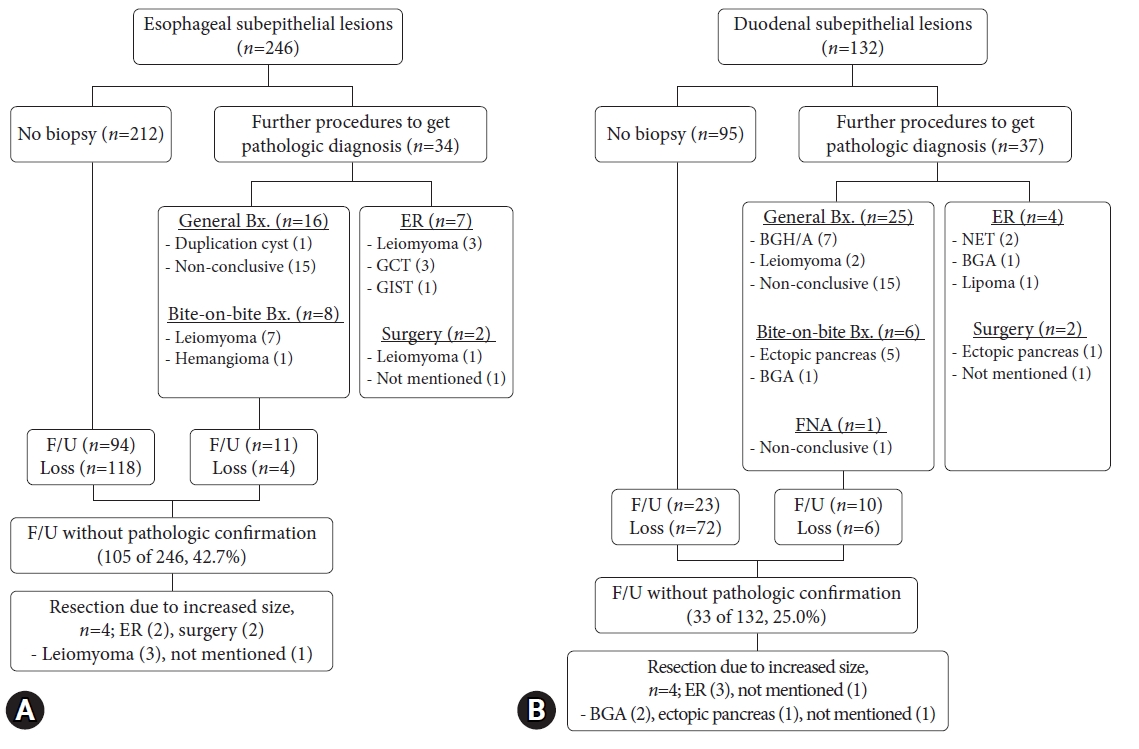
Table┬Ā1.Incidence of subepithelial lesions in each of the eight university hospitals
Table┬Ā2.Demographic characteristics of upper gastrointestinal subepithelial lesions
Values are presented as number (%), mean┬▒standard deviation/median (range), or mean┬▒standard deviation. SEL, subepithelial lesion. a) If there is no mention of the location of the esophagus, within 20 cm of the upper incisor was classified as 'upper esophagus,ŌĆÖ and if more than 30 cm from the upper incisor, it was classified as 'lower esophagus.ŌĆÖ The distance to the upper incisor was classified as 'undocumented' if it was not mentioned. Table┬Ā3.Characteristics of upper gastrointestinal subepithelial lesions on endoscopic ultrasound Table┬Ā4.The frequency order of the subepithelial lesions according to the location of the upper gastrointestinal tract REFERENCES1. Lee M, Min BH, Lee H, et al. Feasibility and diagnostic yield of endoscopic ultrasonography-guided fine needle biopsy with a new core biopsy needle device in patients with gastric subepithelial tumors. Medicine (Baltimore) 2015;94:e1622.
2. Kim GH, Cho YK, Kim EY, et al. Comparison of 22-gauge aspiration needle with 22-gauge biopsy needle in endoscopic ultrasonography-guided subepithelial tumor sampling. Scand J Gastroenterol 2014;49:347ŌĆō354.
3. He G, Wang J, Chen B, et al. Feasibility of endoscopic submucosal dissection for upper gastrointestinal submucosal tumors treatment and value of endoscopic ultrasonography in pre-operation assess and post-operation follow-up: a prospective study of 224 cases in a single medical center. Surg Endosc 2016;30:4206ŌĆō4213.
4. Standards of Practice Committee, Faulx AL, Kothari S, et al. The role of endoscopy in subepithelial lesions of the GI tract. Gastrointest Endosc 2017;85:1117ŌĆō1132.
5. Gao Z, Wang C, Xue Q, et al. The cut-off value of tumor size and appropriate timing of follow-up for management of minimal EUS-suspected gastric gastrointestinal stromal tumors. BMC Gastroenterol 2017;17:8.
6. Ryu DG, Choi CW. Common gastric subepithelial tumors in Koreans. Korean J Helicobacter Up Gastrointest Res 2022;22:29ŌĆō37.
7. Lim YJ, Son HJ, Lee JS, et al. Clinical course of subepithelial lesions detected on upper gastrointestinal endoscopy. World J Gastroenterol 2010;16:439ŌĆō444.
8. Lee JH, Lee HL, Ahn YW, et al. Prevalence of gastric subepithelial tumors in Korea: a single center experience. Korean J Gastroenterol 2015;66:274ŌĆō276.
9. Sharzehi K, Sethi A, Savides T. AGA clinical practice update on management of subepithelial lesions encountered during routine endoscopy: expert review. Clin Gastroenterol Hepatol 2022;20:2435ŌĆō2443.
10. Moon JS. Role of endoscopic ultrasonography in guiding treatment plans for upper gastrointestinal subepithelial tumors. Clin Endosc 2016;49:220ŌĆō225.
11. Kim SM, Kim EY, Cho JW, et al. Predictive factors for differentiating gastrointestinal stromal tumors from leiomyomas based on endoscopic ultrasonography findings in patients with gastric subepithelial tumors: a multicenter retrospective study. Clin Endosc 2021;54:872ŌĆō880.
12. Bang CS, Baik GH, Shin IS, et al. Endoscopic submucosal dissection of gastric subepithelial tumors: a systematic review and meta-analysis. Korean J Intern Med 2016;31:860ŌĆō871.
13. Kim TW, Kim GH, Park DY, et al. Endoscopic resection for duodenal subepithelial tumors: a single-center experience. Surg Endosc 2017;31:1936ŌĆō1946.
14. Deprez PH, Moons LM, O╩╝Toole D, et al. Endoscopic management of subepithelial lesions including neuroendocrine neoplasms: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2022;54:412ŌĆō429.
15. Kim MN, Kang SJ, Kim SG, et al. Prediction of risk of malignancy of gastrointestinal stromal tumors by endoscopic ultrasonography. Gut Liver 2013;7:642ŌĆō647.
16. Kim SE, Park MI. Natural course of gastric subepithelial tumor. Korean J Helicobacter Up Gastrointest Res 2015;15:1ŌĆō8.
17. Cho JW; Korean ESD Study Group. Current guidelines in the management of upper gastrointestinal subepithelial tumors. Clin Endosc 2016;49:235ŌĆō240.
18. Zhang XC, Li QL, Yu YF, et al. Diagnostic efficacy of endoscopic ultrasound-guided needle sampling for upper gastrointestinal subepithelial lesions: a meta-analysis. Surg Endosc 2016;30:2431ŌĆō2441.
19. Pouw RE, Barret M, Biermann K, et al. Endoscopic tissue sampling: Part 1: Upper gastrointestinal and hepatopancreatobiliary tracts. European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2021;53:1174ŌĆō1188.
20. Lee JS, Cho CM, Kwon YH, Seo AN, Bae HI, Han MH. Comparison of diagnostic performances of slow-pull suction and standard suction in endoscopic ultrasound-guided fine needle biopsy for gastrointestinal subepithelial tumors. Clin Endosc 2022;55:637ŌĆō644.
22. Chen H, Li B, Li L, et al. Current status of endoscopic resection of gastric subepithelial tumors. Am J Gastroenterol 2019;114:718ŌĆō725.
23. Lee HL, Kwon OW, Lee KN, et al. Endoscopic histologic diagnosis of gastric GI submucosal tumors via the endoscopic submucosal dissection technique. Gastrointest Endosc 2011;74:693ŌĆō695.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
