INTRODUCTION
Recent advances in medical technology have made endoscopy a preferred and accessible option for the evaluation and treatment of various gastrointestinal lesions. From image-enhanced endoscopy to advanced therapeutic endoscopy, what was once considered a boundary has been continually challenged and expanded. Despite these advances, the biggest limitation of using flexible endoscopes for endoscopic intervention lies in their innate nature. In freely moving gastrointestinal organs with tortuosity, endoscopist manipulations cannot be accurately delivered to the distal tip of the flexible endoscope. Moreover, having only one working channel permits the use of only one device, even in challenging therapeutic procedures.
Cap-assisted endoscopy refers to a procedure in which a short tube made of a polymer (mostly transparent) is attached to the distal tip of the endoscope to enhance its diagnostic and therapeutic capabilities. Attaching caps to endoscopes offers several advantages over conventional endoscopy. It is particularly useful in: (1) minimizing blind spots during screening colonoscopy, (2) providing a constant distance from a lesion for clear visualization during magnifying endoscopy, (3) accurately assessing the size of various gastrointestinal lesions, (4) preventing mucosal injury during foreign body removal, (5) securing adequate workspace in the submucosal space during endoscopic submucosal dissection (ESD) or third space endoscopy, (6) providing an optimal approach angle to a target, and (7) suctioning mucosal and submucosal tissue with negative pressure for resection or approximation.
The length, width, or shape of the cap, as well as its transparency, material, and the presence of draining holes, can differ according to its purpose. Here, we review various applications of attachable caps in diagnostic and therapeutic endoscopy and their future implications.
MINIMIZATION OF BLIND SPOTS VIA ENHANCED MUCOSAL EXPOSURE
Colonoscopy reduces the incidence and mortality rates of colorectal cancer. However, post-colonoscopy colorectal cancer accounts for 7.2% to 9% of all colorectal cancers.1,2 A substantial proportion of premalignant or malignant lesions are overlooked during screening colonoscopy, even by experienced endoscopists.3,4 Several reasons for this have been proposed, including the small size or flatness of the lesions, insufficient withdrawal time, or poor bowel preparation.5-7
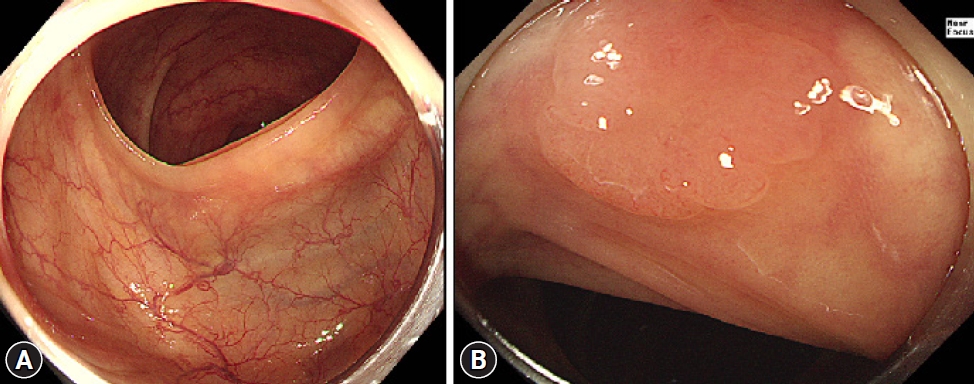
Mucosal exposure is important in improving adenoma detection rates (ADR) and reducing miss rates. Transparent caps are among the most extensively studied distal attachments for improving mucosal exposure. It can press against the colonic folds, thereby improving the view of the mucosa on the oral side of the folds and allowing for the detection of adenomas previously hidden in blind spots.8 It can also help manage the ŌĆ£hookingŌĆØ of the folds, in which the end of the colonoscope will pull back the colonic folds more effectively during the process of straightening the tortuous colonic segment (Fig. 1). This can also result in the exposure of previously uncharted blind spots for further detection of adenomas.
As such, cap-assisted colonoscopy can provide a clear benefit in maximizing ADR, and previous studies have largely agreed. A randomized, two-center trial from 2015 reported an ADR of 42% for cap-assisted colonoscopy, which is higher than the standard ADR of 30% for males and/or 20% for females as proposed by the American Society for Gastrointestinal Endoscopy for quality control.9 A more recent prospective, multicenter, randomized controlled trial showed that cap-assisted colonoscopy, when combined with chromoendoscopy, markedly improved the ADR by 9.5% in comparison to the standard colonoscopy group.10
Earlier analyses reported a marginal benefit of cap-assisted colonoscopy over conventional colonoscopy, both in polyp detection and cecal intubation time,11-13 and recent publications have also shown that the ADR of cap-assisted colonoscopy was significantly higher, especially when trials that did not meet quality standards were excluded.14
PROVISION OF A CONSTANT DISTANCE FROM A LESION FOR CLEAR VISUALIZATION
Magnifying endoscopy, which can be combined with narrow-band imaging or chromoendoscopy, allows more accurate histologic prediction and/or estimation of invasion depth for colorectal neoplasia.15 Today, even a conventional high-definition colonoscope (CF-HQ290; Olympus Co.) provides an optical magnification function, the so-called near-focus mode, which can enlarge images up to 75 times their original size when displayed on a 19-inch monitor without sacrificing image quality.
Acquiring clear images with magnifying endoscopy is occasionally challenging. From the lumens of the intestines to consistent interference from respiratory movements, aortic pulsations, and peristalsis, endoscopists struggle with unfavorable circumstances during examination. Thus, a key factor in magnifying endoscopy is ensuring that magnified images are in focus. To achieve this, the attachment of a black rubber cap at the end of the endoscope can be very helpful (Fig. 2). Attaching a cap at the end of the endoscope allows the endoscopist to fix the distance between the lens and mucosal surface at approximately 2 mm, a point of focus at which good magnification of the endoscopic image can be obtained. Whenever a desired lesion is found, the endoscopist can direct and contact the black rubber cap tip to the mucosal surface while applying zoom mode (Fig. 3).15 Simultaneously, the ŌĆ£red out phenomenon,ŌĆØ which refers to blurred visualization due to direct tissue attachment or suction all the way to the lens, can be minimized.16 The caps are also soft and round, which minimizes damage to the friable mucosa.17 Attaching a cap helps endoscopes stay on focus throughout the exam, allowing endoscopists to quickly and easily obtain images of desired lesions that are on focus.
ACCURATE MEASUREMENT OF COLORECTAL LESIONS
The size of colorectal neoplastic polyps is correlated with colorectal cancer risk; the larger the polyp, the greater the chance of malignancy. It requires shorter follow-up intervals after the removal of adenomas 1 cm in size or greater compared with smaller adenomas. Furthermore, the European Society of Gastrointestinal Endoscopy guidelines recommend different methods of polyp removal depending on the polyp size: cold forceps polypectomy for 1 to 3 mm polyps and cold snare polypectomy for 4 to 9 mm polyps.18 Thus, an accurate measurement of adenoma size can affect the colorectal polyp treatment strategies employed and follow-up after resection.
The most used method of size measurement in daily practice is visual estimation during colonoscopy. However, accurate visual estimation is challenging because of barrel distortion from the fisheye lenses of endoscopes.
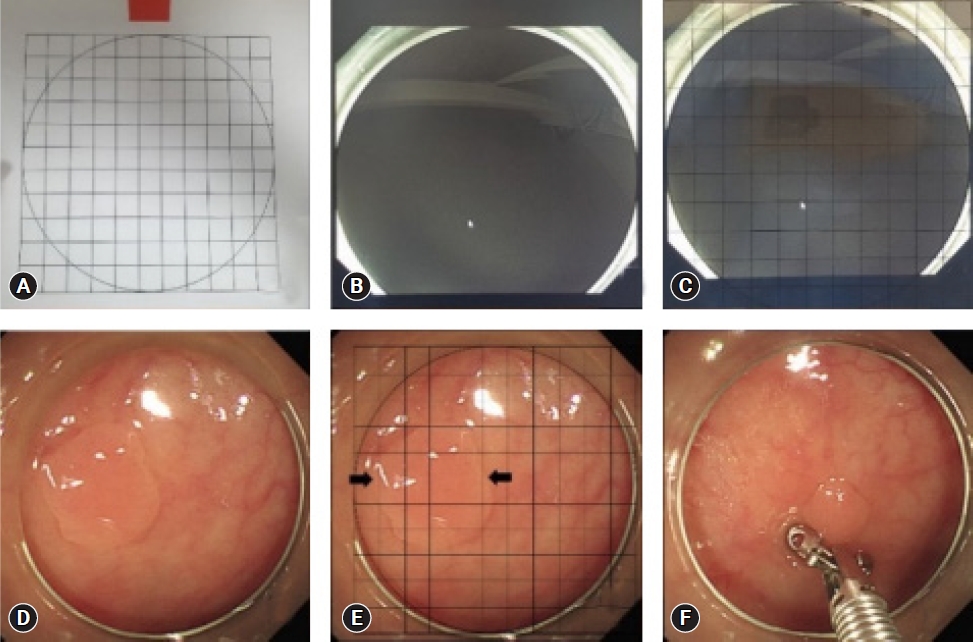
Various add-on tools, such as endoscopic rulers, open forceps, and graduated needles, have been used to improve accuracy in lesion size measurement; however, an add-on colonoscopy cap is the most accessible and affordable option thus far.19-22 In a prospective randomized trial, Han et al.23 placed a transparent grid with 1-mm-spaced grid lines inside a colonoscopic cap upon encountering a polyp to measure its size (Fig. 4). Compared with the control group, measurements taken with colonoscopic caps were significantly more accurate than visual estimation. While measuring with grid paper within the cap prolonged measurement time, the authors suggested that it was still significantly shorter than the time it took endoscopists to use other add-on devices, such as forceps, while providing higher accuracy in measurement than visual estimation.
PROTECTION OF NEARBY MUCOSA DURING FOREIGN BODY REMOVAL
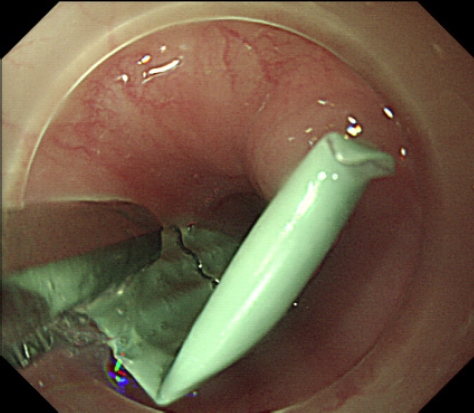
The attachment of an endoscopic cap is effective in protecting the esophageal or pharyngeal mucosa from lacerations that may occur during the retrieval of sharp objects. The endoscopist would grab the sharpest edge of the object with forceps or a grasper, then retrieve it to the confines within the cap to ŌĆ£harborŌĆØ the sharpest edge (Fig. 5). This minimizes the damage that foreign bodies can cause to the normal mucosa during the retrieval process (Fig. 6). In a recent randomized controlled trial in 2021, transparent cap-assisted endoscopy was demonstrated to be a safe and effective method in the management of foreign bodies in the upper esophagus, with a significantly shorter retrieval and operation time, higher technical success and en-bloc removal rates, and a lower rate of procedure-related mucosal tear and bleeding.24 In another propensity score matching study, transparent cap-assisted endoscopy was non-inferior to conventional endoscopy in its success rates for foreign body removal while displaying shorter procedure times and higher rates of clear endoscopic view.25
SECURING ADEQUATE WORKSPACE IN THE SUBMUCOSAL LAYER
In a traditional sense, endoscopy involved two spaces: the gutŌĆōintestinal lumen represented the ŌĆ£firstŌĆØ space and the peritoneal cavity the ŌĆ£second.ŌĆØ However, with advancements in technology, a new arena for endoscopic maneuvers is emerging within the wall of the gut. ŌĆ£Third space endoscopyŌĆØ refers to procedures that take place in the intramural space, a normally enclosed space that must be created by either dissecting or expanding the submucosal layer between the mucosa and muscularis propria (Fig. 7).26,27
In 2007, a new concept of natural orifice transluminal endoscopic surgery was proposed for peritoneoscopy through the stomach.28 This technique was later applied as peroral endoscopic myotomy (POEM) for the treatment of achalasia.29,30 Following the basic concept of the POEM procedure, the tunneling technique for approaching the third space for en-bloc resection of the tumors arising from the muscularis propria was also reported.31 One of the greatest benefits of submucosal tunneling endoscopic resection is that it provides a good workspace within the submucosal layer with a mucosal safety flap valve.
The pocket creation method is a useful method for ESD, which can achieve a higher complete resection rate, higher en-bloc resection rate, shorter procedure time, faster dissection speed, and lower overall adverse event rate than conventional ESD.32,33 This technique can be characterized as entering and dissecting the submucosal layer to make a pocket using a transparent cap after a small mucosal incision, allowing the entrance of the endoscope tip. An attachable cap with a tapered end (Fig. 8) is recommended for this technique34; however, a conventional transparent cap with a normal caliber is also available based on operator preference. The method can yield several advantages over conventional ESD, including prevention of submucosal solution leakage, providing good traction of the submucosal tissue, enabling a tangential approach against the muscularis propria by adjusting the direction of the endoscope inside the pocket, and stable control of the endoscope regardless of heartbeat or respiration.35
PROVIDING AN OPTIMAL APPROACH ANGLE TO A TARGET
The approach angle between the endoscope tip and the target is an important factor in successful endoscopic procedures. However, adjusting the approach angle while managing a flexible endoscope is challenging, even within a freely movable gastrointestinal lumen.
Endoscopic application of hemoclips, called endoclips, is a mechanical hemostatic method used to clamp bleeding vessels; however, their success rate is occasionally dependent on the anatomical location of the lesion. For instance, in lesions on the posterior wall of the gastric body or duodenal papilla, the endoscope is likely to be parallel or extremely tangential to the lesion. Fuke et al.36 reported that of 168 patients with peptic ulcers, 10 of the 88 patients with posterior wall ulcers failed to achieve hemostasis, compared with four of the 80 patients with ulcers in other areas. In that regard, attachable caps can help the endoscope reach a desired lesion more perpendicularly. In their study, Kim et Al.37 showed that while there were no statistically significant differences between the hemostasis rates of patients treated with the cap and those without, placing a transparent cap at the end of the endoscope helped endoscopists clip a lesion too tangential to be clipped with greater ease due to having a superior view. Having a cap also decreased the risk of accidental mucosal damage during clip deployment, as endoscopists would simply cover the clip within the cap prior to its use.
A perpendicular approach to the desired lesion does not always involve the bleeding site in an area with a parallel or tangential angle. In a study comparing cap-assisted endoscopy with conventional endoscopy when visualizing the ampulla of Vater, cap-assisted endoscopy displayed a higher visualization rate, shorter examination time, and higher detection rate of ampullary adenoma.38 In fact, cap-assisted endoscopy was non-inferior in its ability to completely visualize the major duodenal papilla in comparison to side-viewing endoscopy while displaying better scores for mucosal pattern examination and overall satisfaction.39
One of the challenging steps in the placement of a self-expandable metal stent for malignant obstruction is the passage of the guidewire through a lumen-pending obstruction. In this procedure, shortening and unlooping of the colon are important for aligning the axis of the narrowed lumen and working channel of the endoscope. The ŌĆ£red out phenomenonŌĆØ can frequently occur during manipulation because the endoscope lens is likely to touch the surface of the tumor; however, the use of an attachable cap can prevent vision loss during cannulation.
SUCTIONING MUCOSAL AND SUBMUCOSAL TISSUE WITH NEGATIVE PRESSURE FOR RESECTION OR APPROXIMATION
Ligation is one of the most effective hemostatic methods. However, mechanical hemostasis via ligation is challenging, particularly in flat or depressed lesions. In such cases, suctioning the bleeding point, including the adjacent tissue, into an attachable cap with negative pressure can be a good solution for successful ligation. Endoscopic band ligation is one of the most effective methods of bleeding control in variceal bleeding, DieulafoyŌĆÖs lesion, Mallory-Weiss tear, inflammatory polyps, and gastric antral vascular ectasia40; its success is dependent on how ŌĆ£wellŌĆØ the desired lesion is suctioned into the cap opening.
When performing endoscopic mucosal resection (EMR), the use of a cap, commonly referred to as ŌĆ£EMR-C,ŌĆØ is advantageous. During this procedure, the endoscopist presses the cap against the normal mucosa surrounding the target lesion and applies light suction to seal the cap outlet. The snare was then opened and forced to rest along the inner groove of the rim of the cap to form a loop (Fig. 9). The suction was then released, and the cap was used to suck the lesion under a medium to high vacuum. After the endoscopist strangulates the lesion by closing the snare, the suction is again released, allowing the snare to close snugly.41 Without suctioning ability from using caps, it would be difficult to ensnare a desired lesion with ease, and it would also be difficult to resect a lesion larger than the diameter of the endoscope. The cap is also useful in retrieving resected specimens.
An over-the-scope clip (OTSC) (Fig. 10) is another therapeutic tool aided by endoscopic caps. The OTSC was developed to compress tissue in the gastrointestinal tract; by grabbing more tissue, they allow for obliteration of arterial blood flow underneath the stigmata of recent hemorrhage. The OTSC is designed to have a transparent cap at the end of the endoscope, on the outside of which lies a detachable clip that looks like a bear claw. Prior to use, the endoscope, aided by a cap, is placed perpendicularly to position the clip immediately above the target lesion as closely as possible. The lesion is aspirated from the inside of the cap, after which the clip is deployed. The four prongs of the clip anchors the lesion from left to right, continuously compressing the tissue. OTSC is not only used as a last resort in unresolved or recurring GI bleeding, but can also be used in other fields of defects, such as perforations and fistulas. In these cases, an anchor (OTSC Anchor; Ovesco Endoscopy AG) or twin grasper (OTSC Twin Grasper; Ovesco Endoscopy AG) was used to better approximate the tissue. Jensen et al.42 performed a randomized controlled trial that compared OTSCŌĆÖs therapeutic effects as an initial treatment for severe non-variceal upper gastrointestinal bleeding. Their results showed that OTSC significantly reduced the rate of further bleeding, regardless of causes, especially in patients with major stigmata of hemorrhage.
CONCLUSIONS
This review summarizes the key components that make endoscopic cap an attractive accessory for various procedures. Overall, its application may lead to decreased procedure time, better visualization, and fewer procedural complications; simply put, caps maximize the minimally invasive and prompt potential of endoscopes. Nevertheless, endoscopic caps cannot be reimbursed by Korean Health Insurance, which may preclude their appropriate application.