INTRODUCTION
Inflammatory bowel disease (IBD) comprises of two major disease entities: Crohn's disease (CD) and ulcerative colitis. These diseases undergo a relapsing and remitting course. Therefore, determination of inflammatory state is crucial for the assessment of disease activity and for the optimized therapy. However, no simple diagnostic tool for monitoring intestinal inflammation is available.
Among diverse diagnostic imaging modalities, computed tomography (CT) and magnetic resonance (MR) techniques have been optimized, over the last decade, for small bowel imaging with increasing role in the evaluation of small bowel diseases, especially CD. This article introduces the current status of CT enterography (CTE)/MR enterography (MRE) in evaluation of IBD, especially focusing on the assessment of disease activity in CD, and also discusses the current technical issue as well as comparative diagnostic performance.
CTE
In IBD patients, primary role of conventional CT is to evaluate extraenteric manifestation and complications, such as fistulas, abscess, and bowel obstruction. With the improved resolution of multidetector CT, CTE has become an important method of choice for evaluating small bowel diseases; determination of disease activity as well as the extent and severity of inflamed bowel.1,2
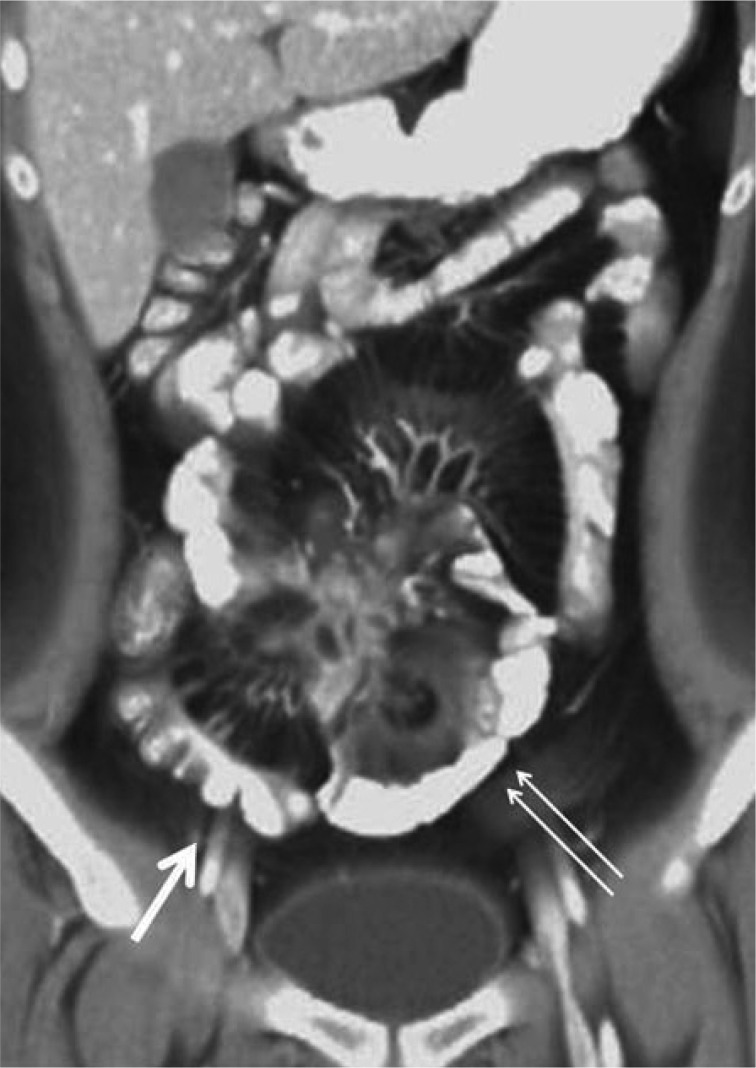
On CTE, CT findings indicative of active inflammation include bowel wall thickening (thickness of >3 mm), mural stratification, mural hyperenhancement, increased attenuation in the perienteric fat, and engorged vasa recta (comb sign)(Fig. 1).1,3
Mural thickening is the most frequently observed CT finding in CD (up to 82% of patients) but not specific to disease activity.4 Mural stratification means the bowel wall layers (a trilaminar pattern) on contrast enhanced CTE by edematous bowel wall thickening. This feature is not specific to CD, however.5 Mural enhancement, most of all, is the most sensitive indicator of disease activity.5 It has also been reported that the degree of bowel wall enhancement correlates with the severity of inflammation.6,7 Increased attenuation of mesenteric fat is often seen in active CD. This CT feature, as well as comb sign, is the most specific sign of disease activity and correlates with the level of C-reactive protein.7 Other CT findings that might be seen in inactive longstanding CD include submucosal fat deposition, sacculation, fibrofatty proliferation, and fibrotic strictures.3
Many researchers have mentioned positive correlation between CTE findings and clinical/biochemical markers of disease activity, such as CD activity index (CDAI), C-reactive protein, and erythrocyte sedimentation rate, respectively, but the clinical relevance of these images is still controversial.7,8
Major negative concern of CTE is the use of ionizing radiation. Many of CD patients are young, and because of their chronic disease, they may have to undergo lifetime repeated imaging examinations. The efforts should be made to reduce radiation dose in these patients by minimizing CT examination, dose reduction (using the dose modulation option or advanced reconstruction techniques), or considering other diagnostic imaging modalities such as MRE.
MRE
Over the last decade, the improved spatial and temporal resolution of MR images, combined with the use of large volumes of oral contrast agents to provide bowel distension, allowed the evaluation of bowel wall contrast enhancement, wall thickening and edema, which are useful findings for the assessment of CD activity.9
With increasing awareness on the risk of radiation exposure, global interest in MR imaging has increased owing to its excellent soft tissue contrast, direct multiplanar imaging capability and lack of ionizing radiation. MRE, these days, plays an important role in the evaluation of small bowel disorders.10 In recent prospective studies, MRE was found to have similar accuracy, area under the receiver operating characteristic curve and sensitivity for detecting active inflammation in CD compared with CTE.11-13 In addition, MRE has the potential advantage of providing functional and quantitative information about bowel wall (e.g., diffusion, perfusion, motility) that cannot be obtained by CT.
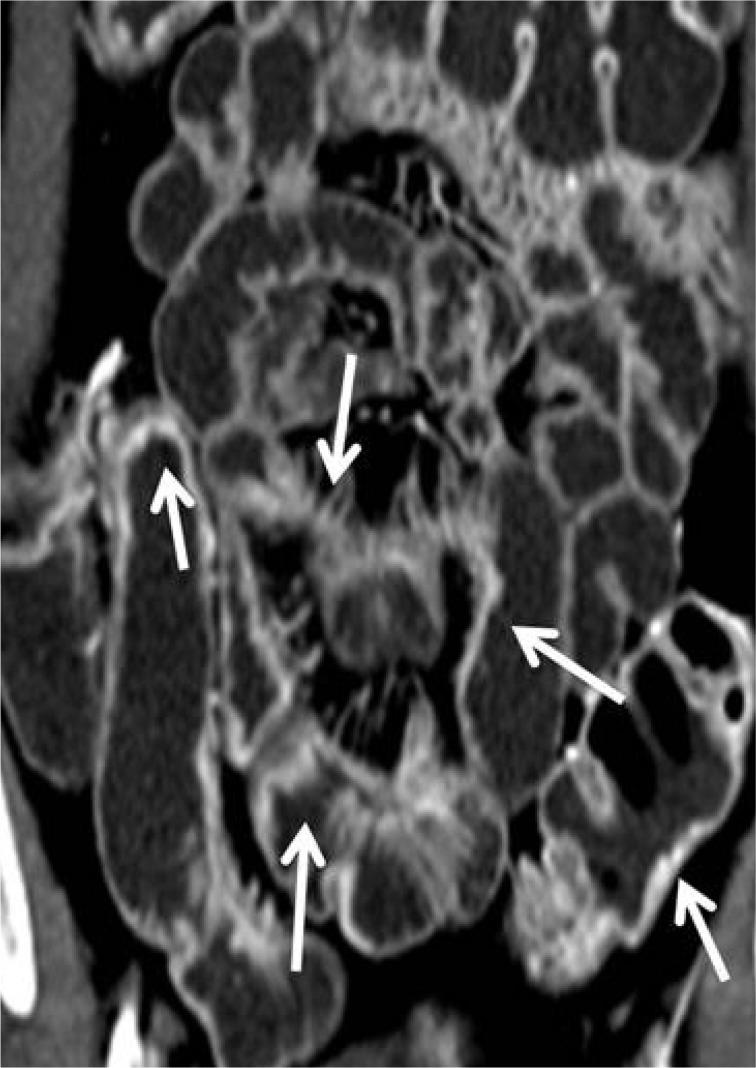
MR findings of actively inflamed small intestine are similar to CT findings indicative of active CD; mural hyperenhancement (Fig. 2), mural thickening (thickness of >3 mm), increased T2 signal in the bowel wall, mural stratification, adjacent fat stranding (streaky decreased signal within the mesenteric fat on non-fat-suppressed T2 weighted images), and "comb sign." Among them, mural thickening, increased enhancement, and increased T2 signal are the most common findings in active CD (77.8% to 100%).14 Sempere et al.15 compared magnetic resonance imaging (MRI) findings of bowel wall thickening and enhancement in patients with active and quiescent CD, and found significant correlation between the degree of enhancement and thickening compared with CDAI. Koh et al.16 reported a sensitivity of 91% and a specificity of 71% for active CD with MRE, whereas using the CDAI the sensitivity was 92% and specificity 28% in the same study.
In some CD patients with strictures, it is important to distinguish between inflammatory stricture and chronic fibrotic stricture, as obstruction and spasm in active CD may be relieved by medical treatment whereas chronic strictures may require surgical intervention. MRE can provide useful information in this setting by differentiating between chronic fibrotic stricture by depicting fat-halo sign (a low signal in submucosal layer produced by fat hypertrophy and fibrosis of the submucosa in chronic IBD) and inflammatory stricture (an intermediate low signal ring produced by submucosal edema).15,17 Fibrotic strictures have been also reported to demonstrate irregular mural enhancement.18
In the evaluation of extramural complications, higher contrast resolution of MRI makes it more suitable for the detection of fistulae and abscesses than CT.19,20 MRE can provide an excellent depiction and staging of perianal fistulas, which are common complications in CD.21-24 Moreover, MRE can be combined with dedicated perianal imaging to provide a comprehensive evaluation of overall disease activity.
Recently, diffusion weighted imaging (DWI) and quantitative dynamic contrast enhanced MRI (DCE-MRI) have been investigated in the assessment of bowel inflammation in CD.14,25-28 Preliminary results from initial studies appear promising despite their study limitations such as small sample size or an imperfect reference standard. According to Oto et al.14 research, DCE-MRI and DWI can provide so reliable quantitative measures of small bowel inflammation that these imaging tools differentiate actively inflamed bowel segments from normal bowel in CD. DWI provides better sensitivity compared with DCE-MRI, and combination of apparent diffusion coefficient and volume transfer constant (Ktrans) parameters for analysis can potentially improve specificity. These results reflect that actively inflamed small bowel segments in CD demonstrate increased perfusion and restricted diffusion. Although these new emerging techniques appear promising, more cumulative data through prospective large series is necessary to establish their diagnostic role in clinical practice.
DIAGNOSTIC PERFORMANCE OF CTE/MRE
Using fluoroscopic examination such as enteroclysis or small bowel follow through (SBFT) as reference standards, the sensitivity of CTE for active CD had been estimated to be over 85%.29,30 Using endoscopic or surgical findings as a reference standard, the sensitivity of CTE and MRE for active inflammation has been estimated to be 77% to 92%.16,31-33 Our data revealed that CTE, MRE, and SBFT are equally accurate for detecting active CD of the small bowel. CTE and MRE were equally as accurate for identification of active CD of the small bowel (sensitivity of CTE vs. MRE, 89% vs. 83%; specificity of CTE vs. MRE, 80% vs. 100%). And also, the sensitivity values of CTE and MRE (100%) for detection of extraenteric complications were significantly higher than those of SBFT (32% to 37%) (p<0.001).11
Prospective comparison of MRE and CTE by Siddiki et al.13 shows relatively same sensitivity in detection of active small bowel CD (90.5% of MRE, 95.2% of CTE). Until now, the available data concerning accuracy of MRE in detecting disease activity is less than CTE. However, although image quality is still better with CT, early results of MRE are encouraging because of a similar sensitivity and diagnostic effectiveness.11,13,34 Motion artifacts from small bowel motility are more severe with MRE,34 but the use of antiperistaltic agent reduces blurring and artifacts related to bowel motility.35
In prospective comparative trials between MRE and capsule endoscopy (CE),33 sensitivities of CE and CTE in detecting active CD show similar results (82% to 83%), but CTE shows higher specificity (89% for CTE, 53% for CE). Umschaden et al.36 addressed that, in 24% of the patients, MRE demonstrated abnormalities not seen at CE, whereas another study detected 70% or more abnormalities with MRE as compared with CE.37
On the other hand, the feasibility of labeled-leukocyte scintigraphy in detecting active inflammation of CD has been evaluated. 99mTc-hexamethylpropyleneamine oxime is a commonly used agent because of its greater availability, better image quality and lower radiation dose.38-41 According to some researches, the sensitivity and specificity of leukocyte-labeled scintigraphy has been reported to range between 76% to 94.7% and 77.8% to 93.3%, respectively.12 Recent studies on 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) in assessing active CD reveals lower specificity values for MRI.42 The usefulness of PET in differentiating between active and indolent CD is still unclear.
CONCLUSIONS
Nowadays, cross-sectional imaging modalities, such as CTE and MRE, are particularly suited to evaluate small bowel diseases. It is well known that CTE and MRE can provide excellent assessment of disease activity as well as the macroscopic features, extramural abnormalities, and complications of small intestine in patients with CD.
Early mucosal lesions (such as aphthoid lesions), however, are not accurately visualized on CT or MRI, making them less suitable as a first-line examination for suspected early disease. Therefore, until now, CTE appears to be more cost-effective in the long-term assessment and follow-up of patients, especially those with established CD. Although MRE is being used more frequently because of the advantage of lack of radiation, some limitation of MRE such as high cost, longer examination time, and slightly inferior spatial resolution than CTE, hardly makes it the initial imaging modality of choice in many adult patients. So far, the preference of MRE versus CTE seems to be geographical and based on expertise and public policy. Emerging functional imaging techniques like DWI or DCE-MRI look promising in assessment and monitoring of disease activity in CD, but further prospective studies are needed.