AbstractWe developed three e-learning systems for endoscopists to acquire the necessary skills to improve the diagnosis of early gastric cancer (EGC) and demonstrated their usefulness using randomized controlled trials. The subjects of the three e-learning systems were ŌĆ£detection,ŌĆØ ŌĆ£characterization,ŌĆØ and ŌĆ£preoperative assessment.ŌĆØ The contents of each e-learning system included ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£obtaining experience.ŌĆØ All e-learning systems proved useful for endoscopists to learn how to diagnose EGC. Lecture videos describing ŌĆ£the techniqueŌĆØ and ŌĆ£the knowledgeŌĆØ can be beneficial. In addition, repeating 100 self-study cases allows learners to gain ŌĆ£experienceŌĆØ and improve their diagnostic skills further. Web-based e-learning systems have more advantages than other teaching methods because the number of participants is unlimited. Histopathological diagnosis is the gold standard for the diagnosis of gastric cancer. Therefore, we developed a comprehensive diagnostic algorithm to standardize the histopathological diagnosis of gastric cancer. Once we have successfully shown that this algorithm is helpful for the accurate histopathological diagnosis of cancer, we will complete a series of e-learning systems designed to assess EGC accurately.
INTRODUCTIONGastric cancer is reported to have the fourth highest mortality rate in the world.1 The stage of gastric cancer at diagnosis is often advanced, except in Japan and Korea. Therefore, the overall 5-year survival rate is <10%.1 However, the prognosis improves in patients diagnosed at an early stage.2 In addition, endoscopic treatment of early gastric cancer (EGC) ensures a patientŌĆÖs quality of life.3 Hence, early diagnosis of gastric cancer is imperative. Although esophagogastroduodenoscopy (EGD) is useful for detecting EGC,4 endoscopists have difficulty learning the ŌĆ£technique,ŌĆØ gaining ŌĆ£knowledge,ŌĆØ and ŌĆ£obtaining experience.ŌĆØ These three topics are essential to enable endoscopists to detect EGC using EGD.5 To address these difficulties, we developed three types of web-based e-learning systems, tested each through randomized controlled trials (RCTs), and demonstrated their usefulness in ensuring correct and precise endoscopic diagnosis of EGC.5-7 In this review, we introduce the basic diagnostic systems employed in e-learning, the outcomes of RCTs testing the usefulness of these three e-learning systems, and our future projects.
THE PRINCIPLES FOR DEVELOPING E-LEARNING SYSTEMS FOR THE ENDOSCOPIC ASSESSMENT OF EGCThe core principles of the e-learning system are that endoscopists should acquire ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experienceŌĆØ for the endoscopic diagnosis of EGC.8 Therefore, we constructed the contents for all e-learning systems imparting ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experience.ŌĆØ
To teach ŌĆ£techniqueŌĆØ and ŌĆ£knowledge,ŌĆØ we employed video lectures. The alternative, ŌĆ£acquiring experienceŌĆØ by e-learning, is challenging. Therefore, we speculated that repeated self-study with 100 consecutive cases (100 cases of repeated self-study) could be useful for accumulating experience similar to that employed in machine learning (Fig. 1).5
BASIC DIAGNOSTIC SYSTEMS AND THEIR PERFORMANCE IN ŌĆ£DETECTION,ŌĆØ ŌĆ£CHARACTERIZATION,ŌĆØ AND ŌĆ£PREOPERATIVE ASSESSMENTŌĆØ EMPLOYED IN THE E-LEARNING SYSTEMSDetection using conventional white-light imagingWe constructed a color plus surface classification system (CSCS) to characterize mucosal lesions detected using conventional white-light imaging (C-WLI) alone.4,9 The criteria according to the CSCS were as follows: (1) presence of a well-demarcated lesion with irregularity in color, and (2) presence of a well-demarcated lesion with an irregular surface. If the target met either criterion, the diagnosis was ŌĆ£cancerous.ŌĆØ When both were absent, the diagnosis was ŌĆ£noncancerousŌĆØ (Fig. 2).8
We reported that when we applied these criteria to clinical practice, the sensitivity was 81.0%, and the specificity was 88.1%.10 This diagnostic system can help endoscopists improve their skills in the early detection of EGC.
Characterization using magnifying endoscopy with narrow-band imagingWe established a vessel plus surface classification system (VSCS) (Fig. 3),11 which has already proven helpful in the accurate assessment of EGC12,13 and in delineating EGCs for curative endoscopic resection.14 The VSCS has been approved through a multi-society consensus as a standardized magnifying endoscopy diagnostic system.15 The criteria were as follows: (1) Presence of an irregular microvascular (MV) pattern with a demarcation line. (2) Presence of an irregular microsurface (MS) pattern with a demarcation line. If the target lesion met either or both criteria, the diagnosis of the lesion was ŌĆ£cancerous.ŌĆØ If both criteria were absent, the diagnosis was ŌĆ£noncancerous.ŌĆØ
We report the diagnostic performance of magnifying endoscopy with narrow-band imaging (M-NBI) with a high-confidence prediction according to the VSCS. The sensitivity was 85.7%, and the specificity was 99.4%.12 The case determined to be a false-negative was a signet-ring cell carcinoma that was pale in color and did not represent either an irregular MV pattern or an irregular MS pattern without a demarcation line. When we excluded the false-negative case that showed a pale superficial mucosal lesion, the sensitivity increased from 85.7% to 100%.12 Hence, when the VSCS is used for diagnosis using M-NBI, M-NBI is deemed an optical biopsy.
Preoperative assessment to select the optimal therapeutic strategy for EGC (endoscopic vs. surgical resection) for predicting submucosal cancerTo determine the indications for endoscopic resection, we need to assess the (1) histological findings (differentiated vs. undifferentiated type), (2) size, (3) depth of invasion (mucosal vs. submucosal invasion), and (4) presence or absence of an ulcer.16
We focused on the endoscopic diagnosis of the depth of invasion by chromoendoscopy, referring to ŌĆ£the non-extension signŌĆØ (NES).17 The NES is a phenomenon characterized by focal thickness and rigidity of the submucosal layer caused by massive submucosal invasion of cancerous tissue. When endoscopic air is insufflated and the stomach is distended, the form of extension differs depending on the depth of the cancer. For example, T1a-T1b1 cancers and noncancerous mucosa are well extended and flattened. However, two distinct patterns are found in the case of T1b2 cancer. (1) The T1b2 submucosal invasive cancer is not flattened and is poorly extended, forming a trapezoid elevation (Fig. 4). (2) The tips of the mucosal folds, which converge onto the trapezoid elevation, become elevated (Fig. 4). If the endoscopic findings show both or either of these findings, the lesion is determined to be NES-positive. If both findings are negative, it is assessed as NES-negative. The sensitivity of NES for predicting a T1b2 cancer was 92.0% (95% confidence interval [CI], 87.0%ŌĆō97.0%), and the specificity was 97.7% (95% CI 96.7%-98.8%).17
OUTCOMES OF RCTS TESTING THE USEFULNESS OF THE E-LEARNING SYSTEMSThe first e-learning system: ŌĆ£detectionŌĆØIn the first step, mucosal lesions suspicious for EGC should be detected using C-WLI, which is commonly available worldwide. Accordingly, the first e-learning system was designed to enable endoscopists to increase the detection of EGC using C-WLI alone.5 The principles and details of this e-learning system are comprehensively reported elsewhere.8
In this study, participants took a pretest and were randomly assigned to either the e-learning group (ELG) or non-e-learning group (NELG). Only the participants assigned to the ELG could perform e-learning. After 2 months, both groups underwent a posttest. Eligibility was determined by 515 endoscopists from 35 countries, and 332 endoscopists were enrolled in the trial. We then randomly assigned 166 participants to either the ELG or NELG. Finally, we analyzed the data obtained from 151 participants who completed e-learning (ELG) and 144 (NELG) who did not have access to e-learning (Fig. 5). The mean improvement rate┬▒standard deviation in the ELG was 1.24┬▒0.26, which was significantly higher than that of the NELG (1.00┬▒0.16) (p<0.001, unpaired t-test) (Table 1).5 Regarding the other analysis, the scores of the ELG were better than those of the NELG, which means that the e-learning system was beneficial regardless of the pretest rating, participantsŌĆÖ expertise, or geographical origin (Table 1).5
The second e-learning system: characterizationAfter detecting a suspicious mucosal lesion by C-WLI, the next process should be ŌĆ£characterization.ŌĆØ M-NBI helps differentiate between cancerous and noncancerous lesions12,13 and reduces the number of biopsies.12
Although VSCS is simple, endoscopists require substantial effort to acquire ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experienceŌĆØ to be accustomed to this system. Because no systematic learning system has proven its usefulness through a well-designed clinical trial, we developed an e-learning system for EGC diagnosis using M-NBI and tested its efficacy using an RCT. Endoscopists from all over Japan participated in this study. After the participants received test 1, they were randomly allocated to either the ELG or NELG. The ELG could learn the M-NBI diagnosis through the e-learning system. After the ELG finished e-learning, both the ELG and NELG took test 2.
A total of 395 endoscopists from 77 facilities who completed test 1 were randomly assigned to the ELG (n=198) or NELG (n=197) (Fig. 6). The change in the score in test 2 of the ELG was 7.4 points, which was significantly higher than that (0.14 points) of the NELG (p<0.001, unpaired t-test) (Fig. 7).6 This trial demonstrated the usefulness of this second e-learning system for improving the diagnosis of EGC by M-NBI.
The third e-learning system: ŌĆ£preoperative diagnosisAfter completing two e-learning systems for ŌĆ£detectionŌĆØ and ŌĆ£characterization,ŌĆØ we were confident that acquiring ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experienceŌĆØ is beneficial to endoscopists to improve the diagnosis of EGC. Teaching ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ by giving lectures using recorded video clips in an e-learning context is not so difficult. However, the ŌĆ£accumulation of experienceŌĆØ is difficult for every endoscopist. The experience of endoscopists in hospitals with few cases is limited. We assumed that for predicting submucosal EGC according to endoscopic findings, ŌĆ£experienceŌĆØ is essential for acquiring the diagnostic ability in addition to learning ŌĆ£the techniqueŌĆØ and gaining ŌĆ£the knowledge.ŌĆØ
Accordingly, in contrast to the two previous RCTs, the purpose of the third study was to investigate whether this ŌĆ£accumulation of experienceŌĆØ is independently affected in improving the ability of the endoscopist to predict submucosal EGC.7 We carried out an RCT that included 423 endoscopists from 93 institutions all over Japan. After the participants took a pretest, they learned the ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ by watching recorded videos and the received posttest 1. After posttest 1, we randomly assigned the participants to either the self-study group (SSG) or non-self-study group (NSSG). Only participants in the SSG could log on to the self-study training system, where they practiced 100 case quizzes. After completing the self-study, participants in the SSG and NSSG received posttest 2. The primary endpoint was the difference in the mean scores of posttest 2 between the SSG and NSSG. The maximum score was 100.
A total of 423 endoscopists who completed the pretest were included in the study. Of these, 415 completed posttest 1 and were randomly allocated to either the SSG (n=208) or NSSG (n=207). Finally, 204 participants from the SSG and 205 from the NSSG were analyzed (Fig. 8). After watching the lecture videos, the mean score increased from 72 points on the pretest to 77 points on posttest 1. The posttest 2 score of the SSG participants was 80 points, which was significantly higher than that (76 points) of the NSSG participants (p<0.001, unpaired t-test) (Fig. 9).7
The third study showed that the ŌĆ£accumulation of experience,ŌĆØ achieved by repeated practice using the 100-case self-study training, consolidated the learning ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ and increased the diagnostic ability of endoscopists of EGC.
DISCUSSION AND FUTURE PLANSIn this article, we introduced three web-based e-learning systems to teach the endoscopic diagnosis of EGC, focusing on ŌĆ£detection,ŌĆØ ŌĆ£characterization,ŌĆØ and ŌĆ£preoperative diagnosis.ŌĆØ We clearly demonstrated that these three e-learning systems are useful as shown by RCTs. Before introducing an e-learning system, we also demonstrated a basic diagnostic system and evidence for each system because an e-learning system should be constructed with concrete diagnostic procedures proven useful in well-designed clinical trials.
The e-learning included three components: ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experience.ŌĆØ The endoscopist can learn ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ by viewing lecture videos; however, it is difficult for the endoscopist to acquire ŌĆ£experience.ŌĆØ Accordingly, we developed a 100-case self-training program. The efficacies of the first and second e-learning systems were assessed after participants, who were allocated to the ELG, studied ŌĆ£technique,ŌĆØ ŌĆ£knowledge,ŌĆØ and ŌĆ£experienceŌĆØ altogether. The third e-learning system trial was designed differently from the previous trials because we speculated that 100-case self-training could increase the learning effect. Accordingly, we randomized the participants after completing the video lectures to independently assess the efficacy of the 100-case self-training program. Interestingly, the video lectures significantly improved the test scores; however, this learning effect did not last long without experience. The participants who completed the 100-case self-training program showed significantly greater improvement in the test scores. These results indicated that acquiring ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ is not enough to achieve improved diagnostic ability and that accumulating ŌĆ£experienceŌĆØ is mandatory.18
Conventional instruction, such as teaching in clinical practice by a senior endoscopist, hands-on training using models or patients, attending lectures, and reading papers or books, is useful for imparting ŌĆ£knowledgeŌĆØ and ŌĆ£technique.ŌĆØ However, the number of learners is limited, and these forms of instruction cannot provide an accumulation of ŌĆ£experience.ŌĆØ As described previously, e-learning offers several advantages over conventional instruction. It can provide ŌĆ£techniqueŌĆØ and ŌĆ£knowledgeŌĆØ by watching video clips that are easy for endoscopists to understand, and repeated training programs can be helpful for endoscopists to accumulate ŌĆ£experience,ŌĆØ which is similar to machine learning. Moreover, the number of learners is not limited, and they can access e-learning whenever and wherever they prefer.
One of the limitations of these trials is that outcome measurement was carried out using test scores on the Internet. We have not yet assessed the improvements in clinical practice after e-learning. We aimed to plan clinical trials to assess the improvement in endoscopic diagnosis in clinical practice after participants completed e-learning.
We will consider investigating the efficacy of an e-learning system to improve the histopathological diagnosis of EGC, as this is the gold standard for endoscopic diagnosis. As previously mentioned, a diagnostic system should first be constructed. To date, no standardized diagnostic systems have been established. We developed a comprehensive algorithm for making a histological diagnosis of gastric cancer (Fig. 10). We tested whether the algorithm can function as a standard diagnostic system for e-learning content. In the near future, we will initiate the RCT by inviting international pathologists (UMIN000044545). If this trial is successfully completed, our series of e-learning systems for improving the endoscopic and histopathological diagnosis of EGC can be finalized.
NOTESFig.┬Ā1.An example of the self-study cases for the diagnosis of 100 cases. (A) One case comprises a set of three slides. The first slide shows one endoscopic photo where one lesion is present. First, the participant should click to choose whether the lesion is cancerous or noncancerous. (B) Immediately after clicking on their choice, an illustration indicating whether the answer is correct or incorrect appears on the second slide. (C) The third slide indicates brief instructions on characterizing the endoscopic findings to diagnose correctly and shows the original endoscopic image again. Adapted from Yao et al. EBioMedicine 2016;9:140ŌĆō147, according to the Creative Commons license.5
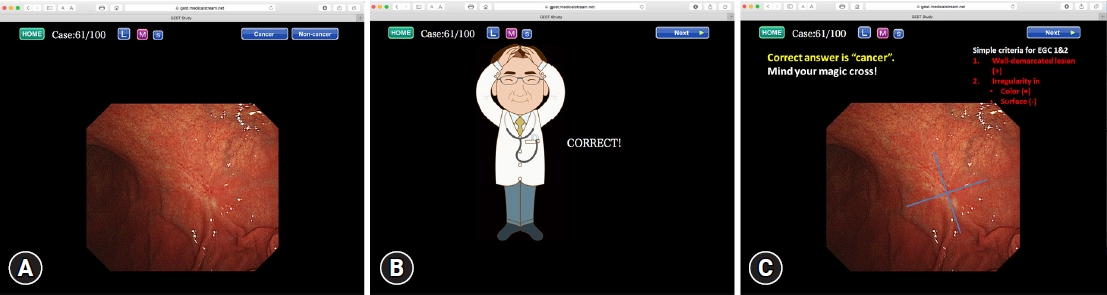
Fig.┬Ā2.Example of endoscopic images showing the differences between noncancerous and cancerous lesions according to the color plus surface classification system (CSCS) by conventional white-light imaging. (A) Endoscopic findings of a focal atrophic pale mucosal lesion. According to the CSCS, the lesion is not well demarcated without irregularity in color or surface. Therefore, this lesion is diagnosed as noncancerous. (B) Endoscopic findings of a pale early mucosal lesion. There is a well-demarcated lesion; the distribution of the color is irregular, and the surface pattern is irregular. Accordingly, this lesion is diagnosed as cancerous. Adapted from Yao et al. Gastric Cancer 2017;20(Suppl 1):28ŌĆō38, according to the Creative Commons license.8
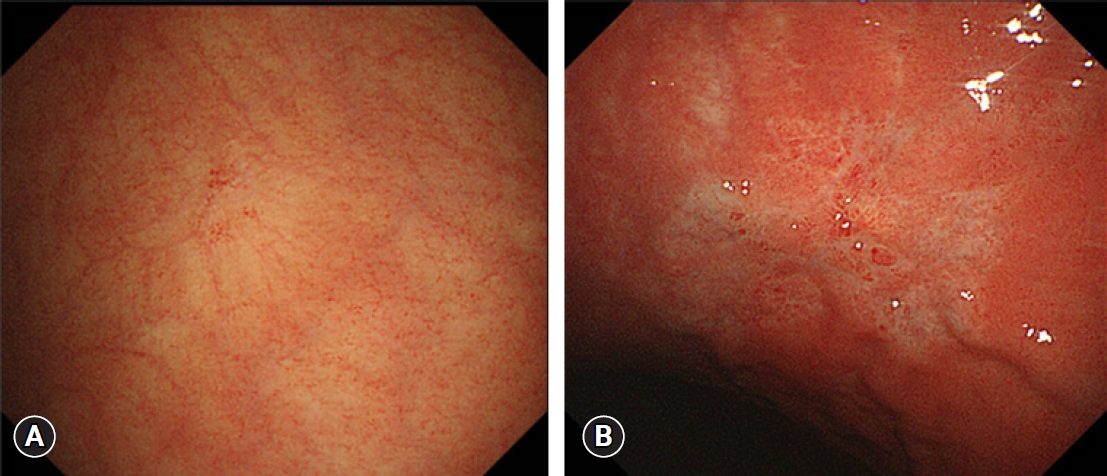
Fig.┬Ā3.Vessel plus surface classification system (VSCS) by magnifying narrow-band imaging. Microvascular and microsurface patterns are classified as regular/irregular/absent. If the findings fulfill the following criteria, a cancer diagnosis is made (arrow, demarcation line): (1) presence of an irregular microvascular (MV) pattern with a demarcation line, and (2) presence of an irregular microsurface (MS) pattern with a demarcation line. Otherwise, the diagnosis is noncancerous. Adapted from Muto et al. Dig Endosc 2016;28:379ŌĆō393, according to the Creative Commons license.15
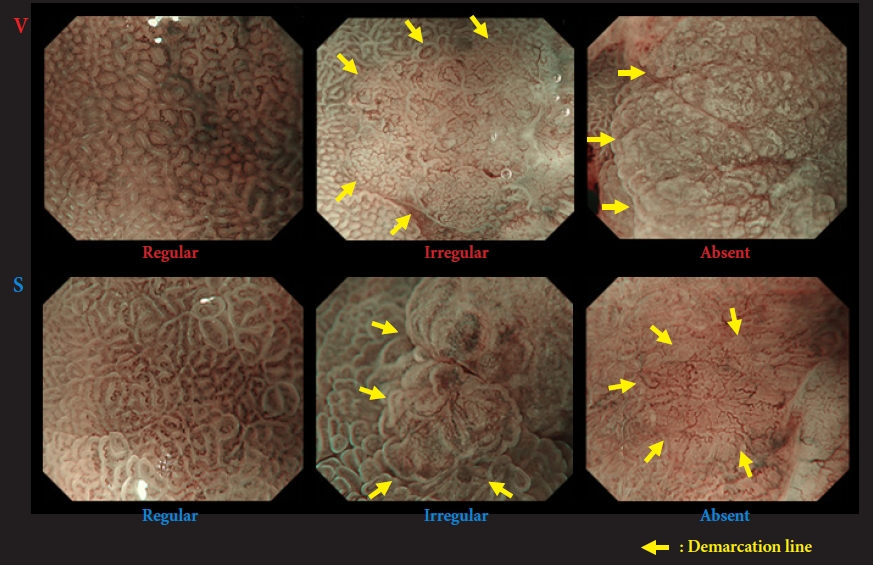
Fig.┬Ā4.A representative endoscopic image of early gastric cancer with deep submucosal invasion (T1b2) showing the non-extension sign. (A) The strongly distended gastric wall is gradually elevated toward the submucosa-invasive area (yellow arrow), which has a trapezoid appearance. (B) This endoscopic finding is well supported by the histological findings of the resected specimen. The depth of invasion is 4,500 ╬╝m from the muscularis mucosae in this case (hematoxylin and eosin staining, ├Ś20). 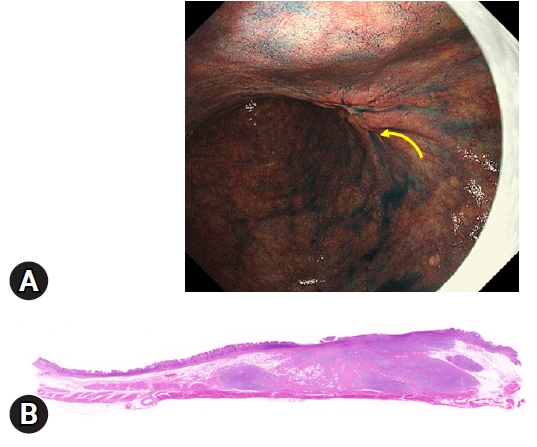
Fig.┬Ā7.The changes in accuracy between tests 1 and 2 in the e-learning and non-e-learning groups. The mean accuracy with 95% confidence intervals in tests 1 and 2 for both groups. The change in accuracy in tests 1 and 2 is significantly higher in the e-learning group than in the non-e-learning group (╬ö7.4 vs. ╬ö0.14 points, respectively; p<0.001, unpaired t-test). Adapted from Nakanishi et al. Endoscopy 2017;49:957ŌĆō967, permission from the publisher.6

Fig.┬Ā8.Flowchart of participantsŌĆÖ enrollment, randomization, and analysis records. The difference with this e-learning study is that to investigate the effect of self-study (accumulating ŌĆ£experienceŌĆØ alone), participants are randomized and allocated to a self-study group or non-self-study group after both groups finish a video lecture showing ŌĆ£techniqueŌĆØ and ŌĆ£knowledge.ŌĆØ 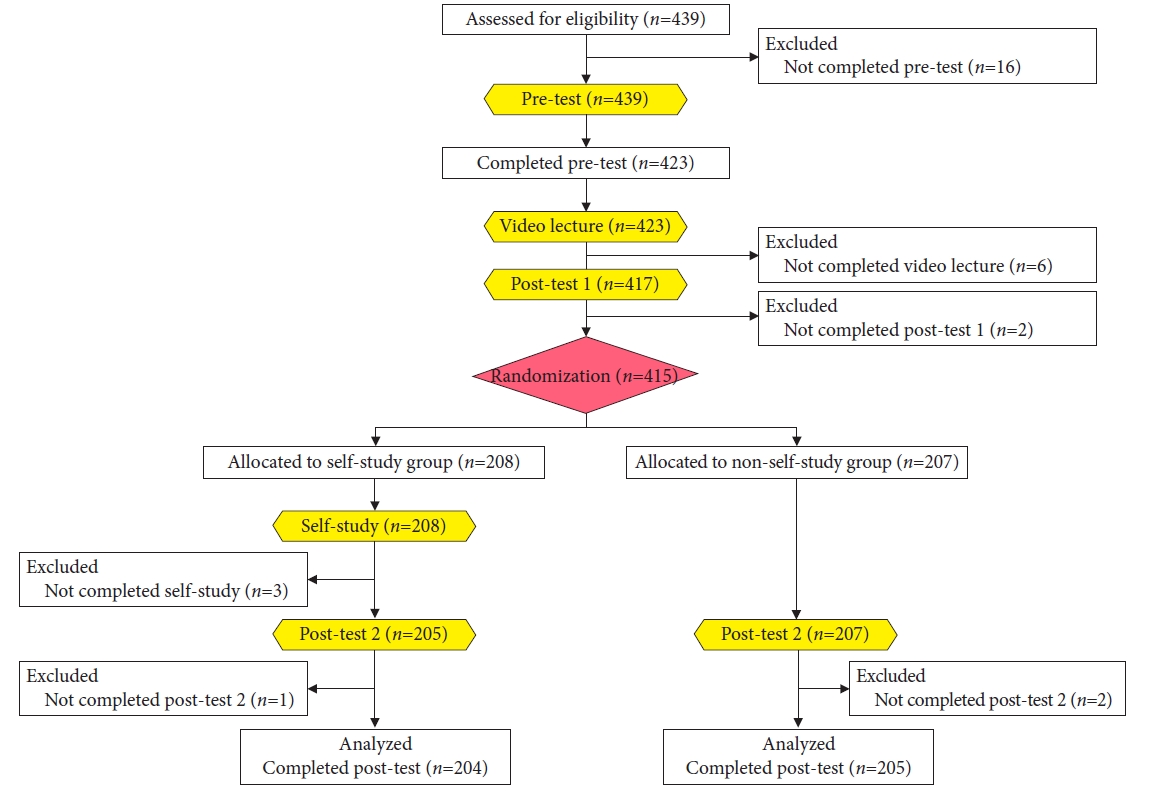
Fig.┬Ā9.Changes in the mean scores with 95% confidence intervals (CIs) for the pretest, posttest 1, and posttest 2. The p values are for comparisons between the two tests. In the self-study and non-self-study groups, the mean scores for posttest 2 (80.3 and 75.8) and posttest 1 (77.4 and 77.1) are significantly higher than those for the pretest (72.1 and 71.4; p<0.001, paired t-test). In the self-study group, the mean score for posttest 2 (80.3) is significantly higher than that for posttest 1 (77.4; p<0.0001, unpaired t-test). In the non-self-study group, the mean score for posttest 2 (75.8) is significantly lower than that for posttest 1 (77.1; p=0.012, paired t-test). Adapted from Kato et al. Endosc Int Open 2019;7:E871ŌĆōE882, according to the Creative Commons license.7
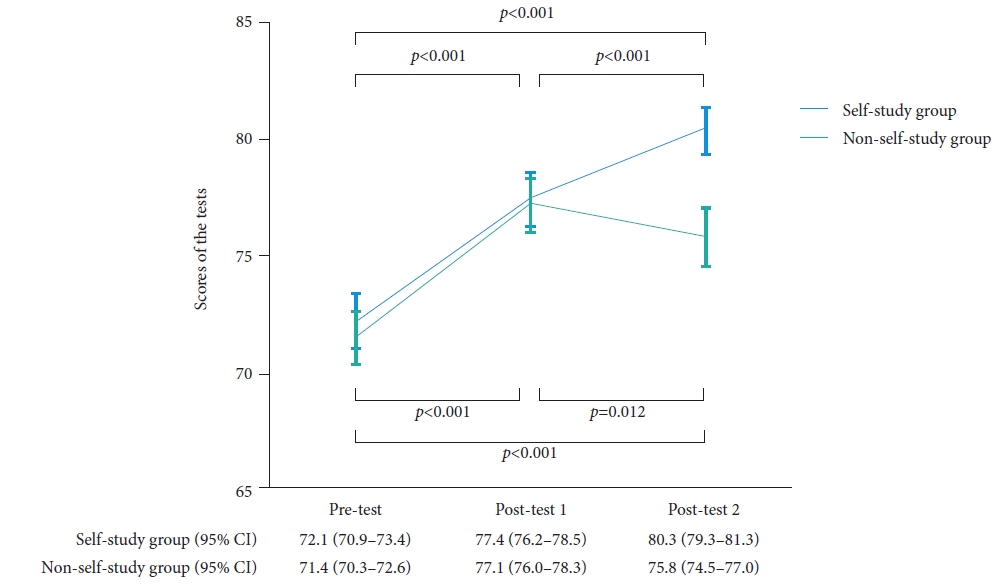
Fig.┬Ā10.The algorithm of the histological diagnostic procedure for gastric epithelial lesions. N/C, nucleus to cytoplasmic; LG, low grade; HG, high grade. a)Except for pyloric gland adenoma. 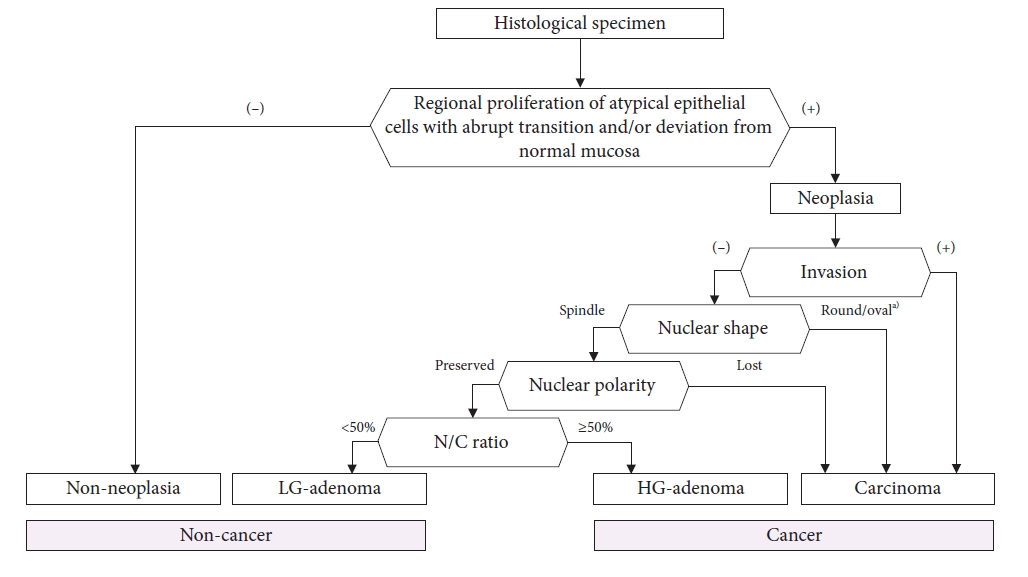
Table┬Ā1.Degree of improvement in test scores between the e-learning and non-e-learning groups
Adapted from Yao et al. EBioMedicine 2016;9:140ŌĆō147, according to the Creative Commons license.5 SD, standard deviation. REFERENCES1. Yang L, Ying X, Liu S, et al. Gastric cancer: Epidemiology, risk factors and prevention strategies. Chin J Cancer Res 2020;32:695ŌĆō704.
2. Hasuike N, Ono H, Boku N, et al. A non-randomized confirmatory trial of an expanded indication for endoscopic submucosal dissection for intestinal-type gastric cancer (cT1a): the Japan Clinical Oncology Group study (JCOG0607). Gastric Cancer 2018;21:114ŌĆō123.
3. Zhang X, Li M, Chen S, et al. Endoscopic screening in Asian countries is associated with reduced gastric cancer mortality: a meta-analysis and systematic review. Gastroenterology 2018;155:347ŌĆō354.
5. Yao K, Uedo N, Muto M, et al. Development of an e-learning system for the endoscopic diagnosis of early gastric cancer: an international multicenter randomized controlled trial. EBioMedicine 2016;9:140ŌĆō147.
6. Nakanishi H, Doyama H, Ishikawa H, et al. Evaluation of an e-learning system for diagnosis of gastric lesions using magnifying narrow-band imaging: a multicenter randomized controlled study. Endoscopy 2017;49:957ŌĆō967.
7. Kato M, Uedo N, Nagahama T, et al. Self-study of the non-extension sign in an e-learning program improves diagnostic accuracy of invasion depth of early gastric cancer. Endosc Int Open 2019;7:E871ŌĆōE882.
8. Yao K, Uedo N, Muto M, et al. Development of an e-learning system for teaching endoscopists how to diagnose early gastric cancer: basic principles for improving early detection. Gastric Cancer 2017;20(Suppl 1):28ŌĆō38.
9. Yao K, Doyama H, Tsuji S. Endoscopic characterization of gastric lesions and resection strategy. In: Testoni PA, Inoue H, Wallace MB, editors. Gastrointestinal and pancreatico-biliary diseases: advanced diagnostic and therapeutic endoscopy. Springer; 2021. p. 151ŌĆō170.
10. Yoshida N, Doyama H, Yano T, et al. Early gastric cancer detection in high-risk patients: a multicentre randomised controlled trial on the effect of second-generation narrow band imaging. Gut 2021;70:67ŌĆō75.
11. Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy 2009;41:462ŌĆō467.
12. Yao K, Uedo N, Kamada T, et al. Guidelines for endoscopic diagnosis of early gastric cancer. Dig Endosc 2020;32:663ŌĆō698.
13. Ezoe Y, Muto M, Uedo N, et al. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology 2011;141:2017ŌĆō2025.
14. Nagahama T, Yao K, Uedo N, et al. Delineation of the extent of early gastric cancer by magnifying narrow-band imaging and chromoendoscopy: a multicenter randomized controlled trial. Endoscopy 2018;50:566ŌĆō576.
15. Muto M, Yao K, Kaise M, et al. Magnifying endoscopy simple diagnostic algorithm for early gastric cancer (MESDA-G). Dig Endosc 2016;28:379ŌĆō393.
16. Yao K, Doyama H, Gotoda T, et al. Diagnostic performance and limitations of magnifying narrow-band imaging in screening endoscopy of early gastric cancer: a prospective multicenter feasibility study. Gastric Cancer 2014;17:669ŌĆō679.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
