INTRODUCTION
Cronkhite-Canada syndrome is a rare gastrointestinal polyposis syndrome with distinctive clinical features and endoscopic findings. Diagnosis can be challenging without suspicion, and the disease carries high mortality due to complications such as infection, gastrointestinal bleeding, and malignancies.
We report two cases of Cronkhite-Canada syndrome that occurred after coronavirus disease 2019 (COVID-19) mRNA vaccination, providing detailed endoscopic images. The institutional review board's approval was waived by our hospital's ethics committee. The study was performed according to the principles of the Declaration of Helsinki, and we obtained the patientsŌĆÖ written consent for this study.
CASE REPORTS
Case 1
A 61-year-old woman presented with chronic watery diarrhea lasting 2 months. Diarrhea occurred 5 to 10 times a day and often contained blood and pus. Periumbilical pain, hypogeusia, anorexia, nausea, dyspepsia, and weight loss accompanied. The symptoms began 3 days after COVID-19 mRNA booster vaccination (Moderna). She had no underlying disease and denied intake of supplements or herbal medications.
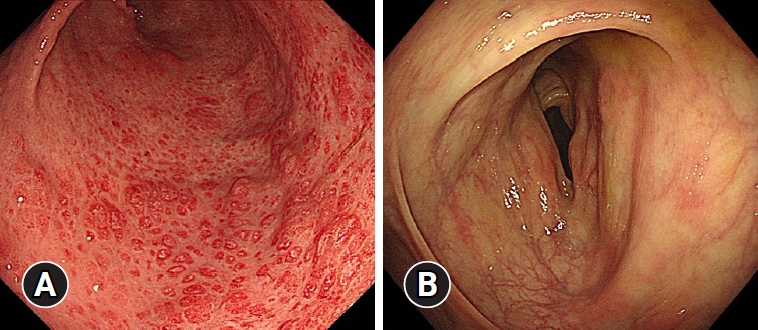
Physical examination revealed a dehydrated tongue, diffuse abdominal tenderness without peritoneal irritation, alopecia, and onychodystrophy (Fig. 1). Blood tests were unremarkable, except for hypoalbuminemia and hypokalemia. Computed tomography revealed diffuse wall thickening of the stomach and colon without enlarged lymph nodes. The esophagogastroduodenoscopy and ileocolonoscopy revealed numerous erythematous polyps in the stomach (Fig. 2A) and colon (Fig. 2B) and atrophied villi in the duodenum (Fig. 2C) and terminal ileum (Fig. 2D). Histology reports of polyps revealed chronic active inflammation with glandular distension, interstitial edema, and a crypt abscess. Helicobacter pylori and cytomegalovirus were negative.
She was diagnosed with Cronkhite-Canada syndrome. As the symptoms did not improve with 10 days of symptomatic treatments, including intravenous nutrition, hydration, and anti-diarrheal medication, a corticosteroid was administered (prednisolone, 30 mg/day), and the symptoms improved in few days. The corticosteroids was tapered over 8 weeks.
Alopecia and onychodystrophy improved. Diarrhea improved and worsened repeatedly, but overall, it improved, and the lost weight was regained. Six months later, she presented no symptoms, and most of the polyps in the stomach and colon disappeared on the follow-up esophagogastroduodenoscopy (Fig. 3A) and ileocolonoscopy (Fig. 3B).
Case 2
A 63-year-old woman presented with chronic watery diarrhea lasting 1 month. Diarrhea occurred more than five times a day and often contained blood and pus. Periumbilical pain, hypogeusia, anorexia, nausea, dyspepsia, and weight loss accompanied. The symptoms began 2 months after COVID-19 mRNA booster vaccination (Pfizer). She had no underlying disease and denied taking supplements or herbal medications.
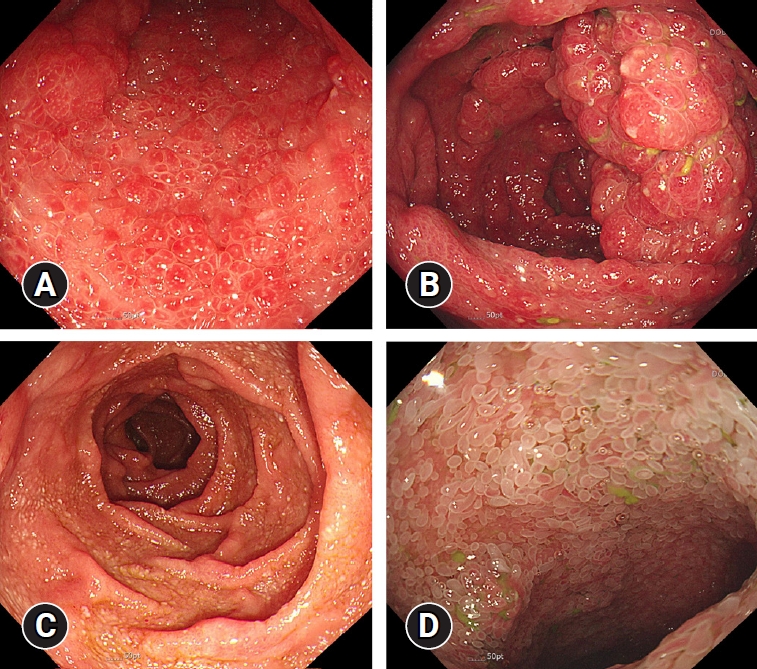
Physical examination revealed a dehydrated tongue, diffuse abdominal tenderness without peritoneal irritation, alopecia, and onychodystrophy (Fig. 4). Blood tests were unremarkable, except for hypoalbuminemia and hypokalemia. Computed tomography displayed diffuse wall thickening of the stomach and colon without enlarged lymph nodes. The esophagogastroduodenoscopy and ileocolonoscopy revealed numerous erythematous polyps in the stomach (Fig. 5A) and colon (Fig. 5B), atrophied villi in the duodenum (Fig. 5C), and edematous villi in the terminal ileum (Fig. 5D). Histology reports of polyps revealed chronic active inflammation with glandular distension and a crypt abscess. H. pylori and cytomegalovirus were negative.
She was diagnosed with Cronkhite-Canada syndrome. As the symptoms did not improved after two weeks of symptomatic treatments, including intravenous nutrition, hydration, and anti-diarrheal medication, a corticosteroid was administered (prednisolone 30 mg/day) and the symptoms immediately improved. The corticosteroid was tapered over 8 weeks without recurrence of symptoms, and the lost weight was regained. However, a follow-up endoscopy was not performed as she did not return to our hospital.
DISCUSSION
Cronkhite-Canada syndrome is a rare disease with hundreds of cases reported worldwide.1 The disease is characterized by hamartomatous polyposis of the gastrointestinal tract, except for the esophagus. The condition typically presents with chronic diarrhea accompanied by hypogeusia, onychodystrophy, alopecia, abdominal discomfort, xerostomia, skin hyperpigmentation, and weight loss. While the etiology is unknown, autoimmune reactions are suggested to contribute to the disease's pathogenesis. Stress, H. pylori infection, and allergic reactions to Chinese alternative medications were reported as possible triggers. There is no standard treatment for the disease. However, corticosteroids, infliximab, azathioprine, and 5-aminosalicylate have been reported to have a positive effect in severe cases.2,3 Gastrointestinal bleeding, malnutrition, and infection are major causes of death, and the patients are at substantial risk for gastrointestinal malignancies.1,4 This paper presents two cases of Cronkhite-Canada syndrome occurring after COVID-19 mRNA vaccination. In both cases, typical clinical findings, such as hypogeusia, onychodystrophy, alopecia, and weight loss were observed. In addition, typical polyposis in the gastrointestinal tract was confirmed through endoscopies. Symptomatic treatments did not improve the symptoms, prompting the administration of corticosteroids, resulting in immediate improvement of symptoms and laboratory test results. The patients improved upon corticosteroids tapering and have not present recurrence of symptoms to date.
There is no established consensus on the pathogenesis of Cronkhite-Canada syndrome. However, immune dysregulation has been implicated because the disease is commonly accompanied by conditions such as hypothyroidism and systemic lupus erythematosus and serology commonly reveals antinuclear antibody positivity.5 Furthermore, case reports suggest the potential association of the disease with IgG4-related disease, due to the favorable response to corticosteroid therapy.6
Even though the exact pathological mechanism remains unknown, previous studies suggest that COVID-19 infection could trigger autoimmunity by inducing rapid autoinflammatory dysregulation in genetic predisposed individuals.7,8 A retrospective study from China reported a 20ŌĆō50% prevalence of autoantibodies in patients with COVID-19 infection.9 The cross-reaction between SARS-CoV-2 proteins and antigens in the human cardiovascular, gastrointestinal, and nervous systems has been suggested as a mechanism by which various autoimmune diseases occur in patients infected with COVID-19.10 There has been a case report of a 40-year-old female diagnosed with Cronkhite-Canada syndrome accompanied by Entamoeba histolytica and COVID-19 infection in Nepal. She reported diffuse abdominal pain, hematochezia, diarrhea, nausea, weight loss, alopecia, onychodystrophy, and hyperpigmentation of the skin, which improved drastically after 4 weeks of corticosteroid therapy. Although the exact mechanism of post-infection Cronkhite-Canada syndrome development is under scrutiny, there have been reports of cases accompanied with infectious diseases that improved with antibiotic treatment. Therefore, the report suggests Cronkhite-Canada syndrome should be suspected in patients with infectious disease if typical symptoms of Cronkhite-Canada syndrome are present.11
Whether the association between the COVID-19 mRNA vaccine and autoimmune diseases is coincidental or causal remains to be elucidated. Increased risk of GuillainŌĆōBarre syndrome with swine influenza vaccination,12 and that of idiopathic thrombocytopenia with the measles-mumps-rubella vaccination13 was confirmed by several studies.14,15 Case reports on new-onset autoimmune diseases, such as immune thrombotic thrombocytopenic purpura, systemic lupus erythematosus, autoimmune liver diseases, and GuillainŌĆōBarre syndrome have also increased after COVID-19 mRNA vaccination.16 A 65-year-old male case of Cronkhite-Canada syndrome after COVID-19 mRNA booster vaccination (Pfizer) was reported in Japan. Alopecia, onychodystrophy, and hypogeusia started 4 weeks after the vaccination. As the result of reverse transcription polymerase chain reaction for COVID-19 was negative, the patient was initially diagnosed with a COVID-19 vaccination-related condition, as the symptoms commonly appeared in patients with COVID-19 infection. Eventually, Cronkhite-Canada syndrome was diagnosed as polyposis, which was observed at colonoscopy, and the symptoms improved after corticosteroid treatment. While symptoms of adverse effects of COVID-19 vaccination are similar to those of Cronkhite-Canada syndrome, the report suggested that if symptoms such as alopecia, hypogeusia, or weight loss occur after COVID-19 vaccination, the possibility of Cronkhite-Canada syndrome should be suspected.17 In our cases report, there is a difference in the timing of symptom onset after COVID-19 mRNA booster vaccination. It is difficult to confirm the exact onset in each patient because the possibility that the disease continued in the subclinical period and worsened after booster vaccination cannot be ruled out.
We report two typical cases of Cronkhite-Canada syndrome and the course of the disease following corticosteroid treatment. Although the casual association between Cronkhite-Canada syndrome and COVID-19 infection or vaccination remains unclear, the cases suggest the possibility that Cronkhite-Canada syndrome may be triggered by COVID-19 mRNA vaccination in genetically susceptible individuals. Therefore, Cronkhite-Canada syndrome must be differentiated in such patients with typical symptoms, considering the excellent response to corticosteroid treatment and the poor prognosis of the disease.