INTRODUCTION
Somatostatinoma was first reported in 1997 by Larsson et al.1 Since then, only a handful of cases have been reported worldwide because of the extremely low incidence of this tumor, estimated to be 1 in 4 million.2 Women were found to have higher incidence than men.3 Most of the cases were symptomatic, with some clinical features of inhibitory symptoms such as diabetes mellitus-like symptoms, steatorrhea, cholelithiaisis, and chronic diarrhea. One of the unusual clinical manifestations was upper gastrointestinal bleeding. Asymptomatic somatostatinoma accounted for approximately 10% of reported cases.4 The most common locations for tumors were the duodenum and pancreas.5 This tumor was malignant in 60% to 70% of cases with tumor size usually less than 5 cm. An association between somatostatinoma and some familial neuroendocrine disorders such as neurofibromatosis and Von Hippel-Lindau syndrome was reported in approximately 7% of cases. Definite diagnoses were made according to tissue histopathology including special immunohistochemical stains. Surgery was the mainstay of treatment for these patients. Adjuvant chemotherapy was not advocated after surgery. Despite slow disease progression, 5-year survival after incomplete resection of the tumor was reported to be 60% to 100%.6 We report a case of somatostatinoma with an unusual location and rare manifestation.
CASE REPORT
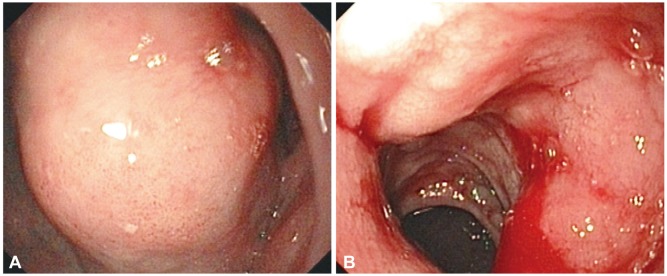
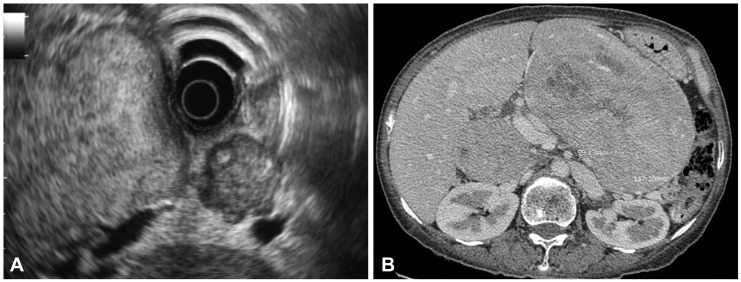
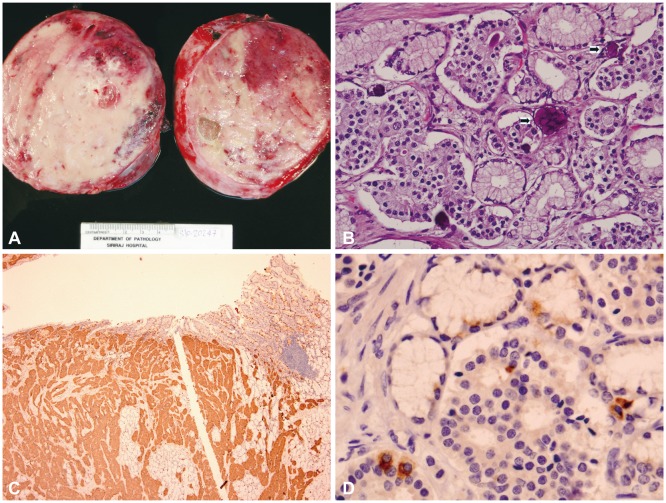
A 49-year-old woman with a history of type 2 diabetes mellitus for a few years presented with chronic abdominal discomfort and nausea for 1 year. She was treated for peptic ulcer disease but did not get better. Three months earlier, she complained of severe abdominal pain, significant weight loss, and chronic intermittent watery diarrhea. Abdominal sonography showed a large epigastric mass, 10У16 cm in size, with multiple hyperechoic nodules at both lobes of the liver and peripancreatic lymphadenopathy, suspected to be metastatic spread to the lymph nodes. Blood test results were within normal limits, and only hyperglycemia was noted. Blood tests for measuring hormone levels were not available in our institute. The patient was scheduled for a computed tomography (CT) scan of the upper abdomen. While she was waiting at the radiology unit, she developed hematemesis and was referred to the internal medicine department for further treatment. She was admitted, and the gastric lavage showed the presence of blood. Her hemoglobin level was 10 mg/dL. Endoscopy showed a large mass, approximately 15 cm in diameter, in the upper gastric body. The mass extended down to the antrum and duodenal bulb with blood oozing (Fig. 1). Next, endoscopic ultrasonography was performed, and a large well-defined isoechoic gastric subepithelial mass more than 13У8 cm in diameter was detected (Fig. 2A). The mass arose from the third and fourth layers of the gastric wall. Multiple intra-abdominal and peripancreatic lymphadenopathy were detected, and the lesions varied in size from 8 to 25 mm. Fine needle aspiration was performed on the mass, and the cytology results led to a suspicion of malignancy. The CT scan showed a large soft tissue mass, with multiple liver metastases and intra-abdominal lymphadenopathy (Fig. 2B). Finally, the patient underwent surgery. Gross pathology showed tumor involvement of the posterior wall of the stomach, with the tumor forming a large intramural multinodular mass (Fig. 3A). Multiple well-circumscribed masses were detected at the pancreatic head and body and at the first part of the duodenum. Multiple liver nodules were also noted. Histopathology showed that the tumor was arranged in solid nests or in an acinar pattern infiltrating from muscularis propria of the stomach through the mucosa and extending to the duodenum. The tumor cells were cuboidal in shape with ample granular eosinophilic cytoplasm. Their nuclei were monotonous and bland, with salt and pepper nuclear chromatin, a typical characteristic of neuroendocrine tumors including positivity for typical characteristic of psammoma bodies (Fig. 3B). Immunohistochemical staining for synaptophysin showed diffuse positivity in the cytoplasm (Fig. 3C). Only a few tumor cells showed scattered positivity for somatostatin on immunohistochemistry (Fig. 3D). A definitive diagnosis of somatostatinoma was made. The patient underwent distal gastrectomy with Billroth I anastomosis and cholecystectomy. However, she developed acute jejunal obstruction 2 weeks later due to intussusception from the jejunal leiomyoma and underwent gastrojejunostomy. She refused any further adjuvant chemotherapy or hormone therapy and received only palliative treatment. She passed away 8 months later because of massive liver metastasis.
DISCUSSION
This case of somatostatinoma involved a tumor, whose size was the largest ever reported. Tumors in most of the reported cases were smaller than 5 cm. More than 85% were symptomatic and more than two-thirds were malignant.7-9 The most common locations of this tumor were the duodenum and pancreas. However, this patient's gastric somatostatinoma was detected in a rare location. In the presented case, the patient only had chronic intermittent watery diarrhea, one of the clinical presentations of classic somatostatinoma syndrome, without any other symptoms. In many other reports, these classic somatostatinoma syndrome presentations were more common for pancreatic masses, whereas obstructive symptoms such as abdominal pain, nausea, and vomiting were more common for duodenal masses. Interestingly, this patient presented with acute massive gastric haemorrhage, which was a rare manifestation of this tumor. Duodenal and periampullary bleeding have been reported in very few cases worldwide as a clinical presentation.10-12 The bleeding was easily stopped with an adrenaline injection. Surprisingly, the endosonographic study could not demonstrate any difference between this tumor and other subepithelial tumors such as gastrointestinal stromal tumor. In our previous study,13 it was very difficult to differentiate the etiology of large subepithelial tumors, especially those larger than 9 cm in diameter, using only endosonographic characteristics. Therefore, a definite diagnosis must be based on cytology and histopathology with special immunohistochemical stains. No data supports the benefit of adjuvant chemotherapy for the treatment of metastatic somatostatinoma, although few small case series reported that the synthetic somatostatin analogue octreotide, at 0.5 mg subcutaneously per day, had some benefits, including reduced diarrhea and other somatostatinoma symptoms, and stabilised tumor growth. Some studies on somatostatinoma reported no benefit from this treatment, which might be from a different subtype of somatostatin produced from the tumor.14,15 The overall 5-year survival for metastatic somatostatinoma was reported at approximately 40%, whereas that for small nonmetastatic lesions is 100%.15