INTRODUCTION
A calcifying fibrous tumor (CFT) is a rare, benign soft tissue tumor, characterized by its unique pathologic findings such as the presence of hyalinized collagenous fibrous tissue with psammomatous or dystrophic calcification and focal lymphoplasmocytic infiltrates [1]. While the location of the tumor may vary, the stomach is the most common (18%) site, followed by the pleura (9.9%), while the colon is an extremely rare site (1.2%) [2]. Most gastrointestinal CFTs are found incidentally during endoscopy or colonoscopy without symptoms. However, using gastroscopy or colonoscopy alone is difficult to differentiate it from other submucosal tumors, which have malignant potential. Endoscopic ultrasonography (EUS) also has difficulties in distinguishing between CFT and other premalignant subepithelial tumors. Therefore, complete resection is the standard strategy for accurate diagnosis and treatment [2]. To date, most resections have been surgical resections. In the case of gastric CFT, resection through endoscopic submucosal dissection (ESD) has been reported in one case [3]. However, there is no report of endoscopic resection of CFT in the colon. Here, we report a case of successful treatment of subepithelial CFT in the large intestine with ESD.
CASE REPORT
A 49-year old woman was referred to our hospital for a small colonic subepithelial tumor (submucosal tumor) detected by screening colonoscopy. Mucosal biopsy specimens from other clinics showed no specific findings. She had no significant medical history or abdominal symptoms. The physical examination and laboratory findings were normal.
A yellowish protruding lesion about 1 cm in diameter was observed in the hepatic flexure on colonoscopy. The surface mucosa was normal, and the lesion showed a hard consistency. The pillow and rolling signs were positive. These findings were suggestive of a subepithelial tumor. (Fig. 1). EUS was performed for the differential diagnosis of a submucosal tumor and visualized a 1-cm sized homogenous hypoechoic lesion arising in the second and third layer of colonic wall, with the first layer preserved (Fig. 2). There was post-acoustic shadowing of a slightly hyperechoic foci inside the lesion (red arrow). Due to the small lesion size, it was difficult to perform EUS-guided fine-needle aspiration (EUS-FNA), and premalignant subepithelial tumors such as neuroendocrine tumors could not be excluded. The patient had psychological anxiety regarding the unclear tumorous lesion. We decided to perform ESD for diagnostic treatment. Hypertonic saline-epinephrine solution was injected into the submucosa to elevate the lesion. A dual knife (KD-650U; Olympus, Tokyo, Japan) was used for dissecting the submucosa. The lesion was completely resected, and no residual lesion remained (Fig. 3). The tumor was well-demarcated and whitish. Deep mucosal injury occurred during the procedure and an oozing visible vessel was observed. Five hemoclips were used for hemostasis, and bleeding was successfully controlled. There were no specific complications after the procedure, and the patient was discharged the next day.
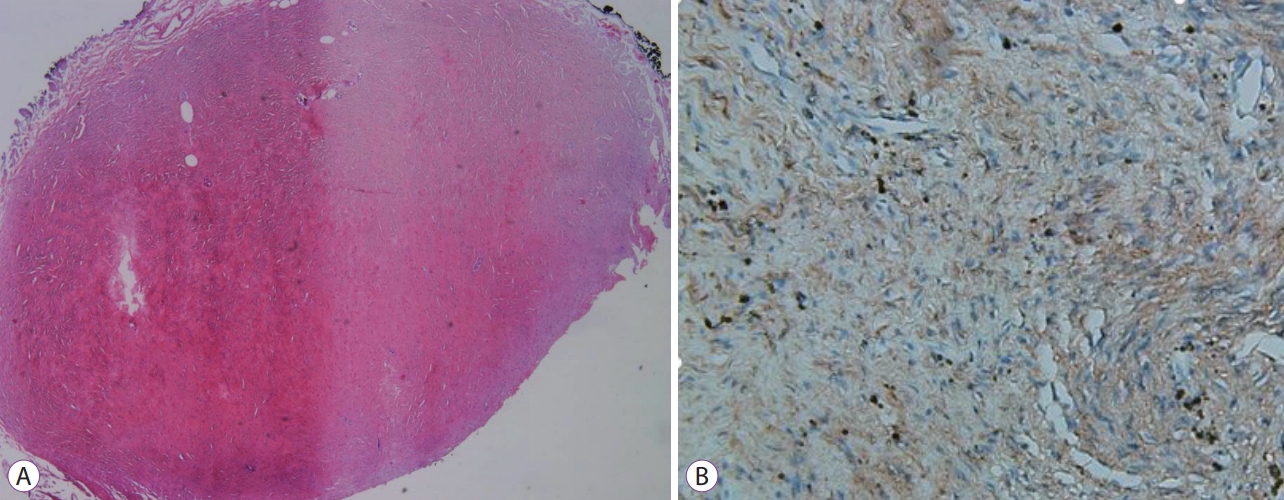
Histologically, a relatively clear, unencapsulated mass was observed. Hematoxylin and eosin stained specimens showed hypocellular spindle cell proliferation, abundant hyalinized collagen, and prominent lymphoplasmacytic inflammatory infiltration. Immunohistochemical staining for Desmin, C-kit, and S100 showed negative results. These histopathologic findings were consistent with CFT (Fig. 4A, B).
DISCUSSION
CFT is a rare benign fibrous tumor that was first reported by Rosenthal et al. [4]. CFT occurs in various parts of the body. The site of disease can encompass a very large area of the body including the stomach (18%), small intestine (8.7%), pleura (9.9%), neck (6.2%), and mesentery (5%) in addition to others, but it is very rarely seen in the large intestine (1.2%) [2]. CFT in the form of a submucosal tumor in the large intestine has been mostly found in the rectum. In the case of intestinal CFT, a submucosal tumor is the most common form, so it is necessary to distinguish it from other unusual tumors (e.g., metastatic tumors or lymphoma) or premalignant submucosal tumors, such as a neuroendocrine tumor [5]. Therefore, in this case, EUS was performed for discrimination, and a 1-cm sized homogenous hypoechoic lesion that occurred in the second layer and third layer was observed on EUS. However, several previous studies reported the unsatisfactory accuracy of EUS (about 43%ŌĆō79%) compared with that of pathologic diagnosis in gastrointestinal submucosal tumors [6,7]. Although an acoustic shadow with calcification was a rare finding in a neuroendocrine tumor, because of the small mass size, accurate diagnosis was difficult. Indeed, there have been reports of metastasis from neuroendocrine tumors less than 1 cm; furthermore, neuroendocrine tumors from the midgut, including the ascending colon, are known to be more likely to have distant metastasis than rectal neuroendocrine tumors [8]. We considered performing pathological evaluation, such as EUS-FNA, for confirmation but the lesion size was about 10 mm and thus, too small to perform EUS-FNA in this case.
Currently, new endoscopic resection techniques such as ESD have been developed and effective endoscopic treatment of subepithelial tumor has become possible. We decided to perform ESD for diagnostic treatment. Through ESD, the lesion was completely resected and histologically confirmed by CFT. Previously, surgical treatment was the standard for submucosal tumors in the gastrointestinal tract. However, in the case of a submucosal tumor, it is more difficult to locate the lesion through endoscopy as in the surgical method, and the complications due to intestinal resection and the risk of surgery itself are often obstacles to the treatment [9].
All colonic CFTs reported so far were confirmed and treated by open surgical resection. Only one case of gastric CFT successfully treated with ESD has been reported [10]. To date, there has been no report of endoscopic treatment of colon CFT, including ESD. The present case is the first report of successful endoscopic diagnosis and treatment of colonic CFT mimicking a submucosal tumor. This case is of significance for showing that a colonic CFT can be well treated with endoscopic procedures. If a submucosal tumor of the colon is found in a suspected case of CFT, ESD may be considered an effective treatment.